The Effect of Amino Acids on Wound Healing: A Systematic Review and Meta-Analysis on Arginine and Glutamine.
Study Design
- Study Type
- Meta-Analysis
- Population
- Patients with wounds
- Intervention
- The Effect of Amino Acids on Wound Healing: A Systematic Review and Meta-Analysis on Arginine and Glutamine. None
- Comparator
- Standard care or placebo
- Primary Outcome
- Wound healing
- Effect Direction
- Positive
- Risk of Bias
- Moderate
Abstract
Under stress conditions, the metabolic demand for nutrients increases, which, if not met, may slow down or indeed stop the wound from healing, thus, becoming chronic wounds. This study aims to perform a systematic review and meta-analysis of the effect of arginine and glutamine supplementation on wound healing. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines were followed for the systematic review and ten electronic databases were used. Five and 39 human studies met the inclusion criteria for arginine and glutamine, respectively. The overall meta-analysis demonstrated a significant effect of arginine supplementation on hydroxyproline content (MD: 4.49, 95% CI: 3.54, 4.45, p < 0.00001). Regarding glutamine supplementation, there was significant effect on nitrogen balance levels (MD: 0.39, 95% CI: 0.21, 0.58, p < 0.0001), IL-6 levels (MD: -5.78, 95% CI: -8.71, -2.86, p = 0.0001), TNFα levels (MD: -8.15, 95% CI: -9.34, -6.96, p < 0.00001), lactulose/mannitol (L/M) ratio (MD: -0.01, 95% CI: -0.02, -0.01, p < 0.00001), patient mortality (OR: 0.48, 95% CI: 0.32, 0.72, p = 0.0004), C-reactive protein (CRP) levels (MD: -1.10, 95% CI: -1.26, -0.93, p < 0.00001) and length of hospital stay (LOS) (MD: -2.65, 95% CI: -3.10, -2.21, p < 0.00001). Regarding T-cell lymphocytes, a slight decrease was observed, although it failed to reach significance (MD: -0.16, 95% CI: -0.33, 0.01, p = 0.07). Conclusion: The wound healing might be enhanced in one or at various stages by nutritional supplementation in the right dose.
TL;DR
The wound healing might be enhanced in one or at various stages by nutritional supplementation in the right dose by nutritional supplemented with arginine and glutamine.
Full Text
nutrients
Review
The Effect of Amino Acids on Wound Healing: A Systematic Review and Meta-Analysis on Arginine and Glutamine
Elena Arribas-López 1, Nazanin Zand 1,*, Omorogieva Ojo 2 , Martin John Snowden 1 and Tony Kochhar 3
- 1 School of Science, Medway Campus, University of Greenwich, Central Ave, Gillingham, Chatham Maritime, Kent ME4 4TB, UK; [email protected] (E.A.-L.); [email protected] (M.J.S.)
- 2 School of Health Sciences, Avery Hill Campus, University of Greenwich, Avery Hill Road, London SE9 2UG, UK; [email protected]
- 3 HCA London Bridge Hospital, Tooley Street, London SE1 2PR, UK; [email protected]
* Correspondence: [email protected]; Tel.: +44-(0)-208-331-8225; Fax: +44-(0)-208-331-8305
Citation: Arribas-López, E.; Zand, N.; Ojo, O.; Snowden, M.J.; Kochhar, T. The Effect of Amino Acids on Wound Healing: A Systematic Review and Meta-Analysis on Arginine and Glutamine. Nutrients 2021, 13, 2498. https://doi.org/10.3390/nu13082498
Academic Editor: Roberto Iacone
Received: 24 June 2021 Accepted: 20 July 2021 Published: 22 July 2021
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Copyright: © 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https:// creativecommons.org/licenses/by/ 4.0/).
Abstract: Under stress conditions, the metabolic demand for nutrients increases, which, if not met, may slow down or indeed stop the wound from healing, thus, becoming chronic wounds. This study aims to perform a systematic review and meta-analysis of the effect of arginine and glutamine supplementation on wound healing. PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines were followed for the systematic review and ten electronic databases were used. Five and 39 human studies met the inclusion criteria for arginine and glutamine, respectively. The overall meta-analysis demonstrated a significant effect of arginine supplementation on hydroxyproline content (MD: 4.49, 95% CI: 3.54, 4.45, p < 0.00001). Regarding glutamine supplementation, there was significant effect on nitrogen balance levels (MD: 0.39, 95% CI: 0.21, 0.58, p < 0.0001), IL-6 levels (MD: −5.78, 95% CI: −8.71, −2.86, p = 0.0001), TNFα levels (MD: −8.15, 95% CI: −9.34, −6.96, p < 0.00001), lactulose/mannitol (L/M) ratio (MD: −0.01, 95% CI: −0.02, −0.01, p < 0.00001), patient mortality (OR: 0.48, 95% CI: 0.32, 0.72, p = 0.0004), C-reactive protein (CRP) levels (MD: −1.10, 95% CI: −1.26, −0.93, p < 0.00001) and length of hospital stay (LOS) (MD: −2.65, 95% CI: −3.10, −2.21, p < 0.00001). Regarding T-cell lymphocytes, a slight decrease was observed, although it failed to reach significance (MD: −0.16, 95% CI: −0.33, 0.01, p = 0.07). Conclusion: The wound healing might be enhanced in one or at various stages by nutritional supplementation in the right dose.
Keywords: arginine; collagen deposition; contraction; food; glutamine; growth factor; gut permeability; interleukin; re-epithelialization; wound healing
1. Introduction
A wound is known as the disruption in the physical continuity of functional tissues [1–4]. The healing process begins immediately after an injury [5–8] and involves four phases [3,9–13]. The healing process consists of a series of sequential and overlapping physiological phases or stages that can persist for years [9,14–17], as shown in Figure 1. It is not a linear process and, depending on diverse extrinsic and intrinsic factors, such as growth factors and cytokines, it can progress both backward and forward through the stages.
1.1. Nutrition
Nutrition is recognized as a key factor in wound healing. Under conditions of stress such as trauma or after surgery, the nutritional demand is increased [18–20] in part due to cell proliferation and protein synthesis [21].
Arginine is a conditionally essential amino acid that is synthesized from citrulline in healthy humans [22] (Figure 2). Based on previous reviews, arginine has been shown to modulate the immune function, hormone secretion, and endothelial function as well as being a precursor to the synthesis of proline in animal and human trials [23–25].
Nutrients 2021, 13, 2498. https://doi.org/10.3390/nu13082498 https://www.mdpi.com/journal/nutrients
Nutrients 2021, 13, 2498 2 of 26
Nutrients 2021, 13, x FOR PEER REVIEW 2 of 26
- Figure 1. Stages of skin wound healing (hemostasis, inflammation, proliferation, and repair and remodeling) over time.
- Figure 2. Metabolism of L-Arginine to produce NO and metabolites involved in the wound healing process. Arginine can be catabolized via the NO synthase pathway. Here, L-Arginine can be converted to L-ornithine and urea by arginase I. Then, by the action of ornithine aminotransferase, ornithine is transformed into proline, which is needed for collagen synthesis. L-ornithine can also be converted to polyamines, which are required for cell proliferation by ornithine decarboxylase [22].
- Figure 1. Stages of skin wound healing (hemostasis, inflammation, proliferation, and repair and remodeling) over time.
- Figure 2. Metabolism of L-Arginine to produce NO and metabolites involved in the wound healing process. Arginine can be catabolized via the NO synthase pathway. Here, L-Arginine can be converted to L-ornithine and urea by arginase I. Then, by the action of ornithine aminotransferase, ornithine is transformed into proline, which is needed for collagen synthesis. L-ornithine can also be converted to polyamines, which are required for cell proliferation by ornithine decarboxylase [22].
There are two pathways in wound healing involving arginine (a) the arginase pathway, which produces polyamines, as well as ornithine and proline. Polyamines are needed for cell proliferation, while the latter ones are required for the synthesis of collagen; and (b) the inducible nitric oxide (NO) synthetase or iNOS pathway, which is a precursor of nitric oxide (Figure 2). NO plays a key role in wound healing as it regulates cell proliferation, collagen formation, and wound contraction [22,26].
Glutamine is the most abundant amino acid found in human blood plasma. It is used as a source of energy for the cells to proliferate, including lymphocytes, macrophages, fibroblasts, and epithelial cells [21,23]. Similar to arginine, its concentration in plasma decreases under conditions of metabolic stress, such as injury, and its depletion is proportional to the acuteness of the trauma [27–30].
There are two pathways in wound healing involving arginine (a) the arginase pathway, which produces polyamines, as well as ornithine and proline. Polyamines are
fibroblasts, and epithelial cells [21,23]. Similar to arginine, its concentration in plasma de-
Some studies have demonstrated that glutamine enhances wound healing, in part, because it increases the concentration of arginine and citrulline, a precursor of arginine (Figure 3). Glutamine thus allows the production of NO in the absence of extracellular arginine in monocytes and macrophages [28,31–34]. This amino acid also reduces gut atrophy [35–39] and limits intestinal permeability [40–46], indirectly reducing the production of proinflammatory cytokines [47]. The intestinal permeability, measured by the lactulose/mannitol excretion ratio, is an important parameter in wound healing since its increment is correlated with the production of proinflammatory cytokines, such as interleukin-6 (IL-6) [48] which plays a key role in the modulation of healing through the regulation of differentiation, activation, and proliferation of keratinocytes, leukocytes, fibroblasts and endothelial cells [49]. Glutamine has also been shown to reduce C-reactive protein (CRP). CRP plays important roles in inflammatory processes and hosts reactions against infections, including NO release, apoptosis, and the production of IL-6 and tumor necrosis factor-α (TNFα). Therefore, CRP levels increase in sick patients and are correlated with the severity of the illness in the patient, thus, objectively quantifying the patient’s stress and acuity [50,51]. Hence, the decline of CRP indicates the reduction of the overall inflammation. Glutamine also acts as an antioxidant through the production of glutathione (GSH) [29,52].
Some studies have demonstrated that glutamine enhances wound healing, in part, because it increases the concentration of arginine and citrulline, a precursor of arginine (Figure 3). Glutamine thus allows the production of NO in the absence of extracellular arginine in monocytes and macrophages [28,31–34]. This amino acid also reduces gut atrophy [35–39] and limits intestinal permeability [40–46], indirectly reducing the production of proinflammatory cytokines [47]. The intestinal permeability, measured by the lactulose/mannitol excretion ratio, is an important parameter in wound healing since its increment is correlated with the production of proinflammatory cytokines, such as interleukin-6 (IL-6) [48] which plays a key role in the modulation of healing through the regulation of differentiation, activation, and proliferation of keratinocytes, leukocytes, fibroblasts and endothelial cells [49]. Glutamine has also been shown to reduce C-reactive protein (CRP). CRP plays important roles in inflammatory processes and hosts reactions against infections, including NO release, apoptosis, and the production of IL-6 and tumor necrosis factor-α (TNFα). Therefore, CRP levels increase in sick patients and are correlated with the severity of the illness in the patient, thus, objectively quantifying the patient’s stress and acuity [50,51]. Hence, the decline of CRP indicates the reduction of the overall inflammation. Glutamine also acts as an antioxidant through the production of glutathione (GSH) [29,52].
Figure 3. Metabolism of glutamine to arginine in human macrophages. Carbamoyl phosphate when combined with ornithine via OTC is converted to citrulline. Then citrulline is transformed into argininosuccinate and then into arginine by the action of ASS and ASL, respectively. Arginine can then be turned into nitric oxide or ornithine. Ornithine can be transformed into glutamine, and vice versa, via glutamate and pyrroline-5-carboxylate.
- 1.2. Why It Is Important to Do This Review
1.2. Why It Is Important to Do This Review
Both arginine and glutamine are considered conditionally essential amino acids. Therefore, they are needed in stress conditions and thus, for wound healing to occur. There is existing information suggesting their mechanism of action in the different stages of healing. However, it is our belief that no previous systematic review and meta-analysis has been conducted on arginine supplementation and its effect on wound healing. In the case of glutamine, its beneficial effect on hospital stay has been reported by two meta-analyses performed by Bollhalder et al. [53] and Novak et al. [54]. However, the first review only included enteral supplementation and focused on the length of hospital stay and mortality. Whereas the second one focused on surgery and critically ill patients. To our knowledge, there is currently no evidence of the effect of their supplementation on different variables affecting wound healing.
Both arginine and glutamine are considered conditionally essential amino acids. Therefore, they are needed in stress conditions and thus, for wound healing to occur. There is existing information suggesting their mechanism of action in the different stages
This systematic review and meta-analysis aimed to evaluate the effect of supplementation of arginine or glutamine on wound healing or parameters related to healing.
2. Materials and Methods
The following systematic review was performed according to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement [55]. Furthermore, this review follows the Population, Intervention, Comparison, and Outcome (PICO) characterization.
2.1. Search Strategy
The databases searched for relevant papers published before October 2020 were Pubmed, American Physiological Society Publications, Taylor & Francis Online, Web of Science, EMBASE, grey literature research with Google Scholar.
Based on the search strategy, the following keywords and synonyms/medical subject headings were used: arginine or glutamine and (inflammation or healing or wound or surgery or cytokines or interleukin or nutrition or hospital stay or C-reactive protein). Words were combined using Boolean operators (OR/AND) (Table 1). References from pertinent articles were also examined for additional studies. Searches were conducted and data from the selected articles were extracted by one researcher (E.A.L.) and cross-checked by another researcher (NZ). For the meta-analysis data, the authors of the selected articles were contacted for the original data when needed.
Table 1. Search terms and search strategy.
Combining Search Terms Patients Amino acid
Patient/Population Intervention Outcome Study Designs
Randomized controlled trial Patients undergoing Surgery OR patients with Pressure Ulcers OR Patients with Critical Illness
Inflammation OR Healing OR Wound OR Cytokines OR Interleukin OR Hospital Stay OR C-reactive protein
Clinical trial OR Randomised controlled trial OR controlled clinical trial
- Column 1 AND
- Column 2 AND
- Column 3 AND Column 4
Amino Acid OR Arginine OR Glutamine OR Nutrition
2.2. Study Selection
Inclusion criteria: studies selected were randomised controlled trials (RCTs) where patients above 18 years old, healthy or not, suffering from acute or chronic wounds were supplemented with arginine or glutamine (Figure 4 and Figure 5).
Exclusion criteria: Studies that did not entail in vivo human studies involving supplementation with arginine or glutamine were excluded from the review. Studies involving participants below 18 years of age were excluded from the review due to the metabolic stress already occurring resulting from growth. Studies involving patients with diabetes, where data were not complete or the original data were not presented and studies in another language other than English, Spanish or French were excluded from the review.
- 2.2.1. Population Adults above 18 years old, healthy or not, suffering from acute or chronic wounds.
- 2.2.2. Intervention Diet supplemented with either arginine or glutamine for at least 5 days.
- 2.2.3. Comparator A control group, either treated with a placebo or not treated.
Nutrients 2021 13, x FOR PEER REVIEW 6 of 26
Nutrients 2021, 13, 2498 5 of 26
- Figure 4. Flow diagram of the search strategy for arginine.
- Figure 5.Figure 5. Flow diagram of the search strategy for glutamine.Flow diagram of the search strategy for glutamine.
- 2.2.4. Outcomes
- 2.3. Data Extraction and Management
- 2.4. Quality Assessment
- 2.5. Data Analysis
Changes from baseline for the intervention were compared with the control in all the parameters analyzed [57]. The pooling of the data was conducted with the metaanalytic methodology, utilizing Cochrane Review Manager 5.4.1 (2020) [58] for the different outcomes evaluated applying fixed effects, the mean differences (MDs) and odds ratio as a degree of effect extent. Nevertheless, for nitrogen balance and T-cell lymphocytes levels, data were converted into standardized mean difference (SMD) owing to the use of different measurement scales. Pooled effect size estimates are presented with their 95% confidence intervals (95% CI). When studies reported multiple results (i.e., multiple-dose), these were included in the meta-analysis as independent comparisons. Heterogeneity was assessed using I2 and Chi2 and considered significant when I2 > 50%. Results were considered significant when the p-value was below 0.05.
3. Results
Five (5) and 39 studies on arginine and glutamine, respectively were included in the systematic review (Figures 4 and 5) (Tables 2 and 3). 3.1. Assessment of Risk of Bias of Included Studies 3.1.1. Risk of Bias of Included Studies on Arginine
The risks of bias in the included studies are shown in Figure 6. 100% of the studies showed a low risk of bias in relation to the random sequence generation and allocation concealment. While less than 75% have demonstrated a low risk of bias with respect to blinding of participants and personnel. In terms of blinding of outcome assessment, incomplete outcome data and selective reporting, all the studies showed a low risk of bias except for Barbul et al. [59] which exhibited an unclear risk of bias. Regarding other risks of bias, less than 50% of the studies showed a low risk of bias whereas the Langkamp-Henken study [60] demonstrated a high risk of bias.
Nutrients 2021, 13, 2498 except for Barbul et al. [59] which exhibited an unclear risk of bias. Regarding other risks7 of 26 of bias, less than 50% of the studies showed a low risk of bias whereas the Langkamp-
Figure 6. Risk of bias summary for the included studies on arginine. Low risk of bias (+), unclear risk of bias (?), and high risk of bias (−).
- 3.1.2. Risk of Bias of Included Studies on Glutamine
- 3.2. Effects of Interventions
- 3.3. Arginine (Arg)
3.1.2. Risk of Bias of Included Studies on Glutamine
The risks of bias in the included studies are shown in Figure 7. Of the studies, 100% showed a low risk of bias in relation to the random sequence generation. All the studies have demonstrated a low or unclear risk of bias regarding blinding of participants except the Goeters et al. [61] and Xian-Li et al. [62] studies, which exhibited a high risk of bias. On the other hand, more than 50% of the studies showed an unclear and high risk of bias in terms of other biases. The high bias is the case of Engel et al. [63] and Goeters et al. studies [61]. With respect to the other risk of bias, more than 75% of the studies exhibited a low risk of bias regarding allocation concealment, blinding of outcome assessment, incomplete data and selective reporting.
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
Nutrients 2021, 13, x FOR PEER REVIEW 8 of 26
Figure 7. Risk of bias summary for the included studies on glutamine. Low risk of bias (+), unclear risk of bias (?), and high risk of bias (−).
Studies evaluating the effect of arginine supplementation on wound healing and reported outcomes.
Barbul et al. [59] mented
Nutrients 2021, 13, 2498 9 of 26
mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
Control Group
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on
Wound-breaking strength,
Barbul et al. Not supple-
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported
↑
3.3. Arginine (Arg)
Nussbaum [64]
Not supplemented
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported outcomes.
↑ Wound-breaking strength, ↑ Lymphocyte mitogenesis Nussbaum Not supple-
Study Duration Patient Population n Dosage Control Group Barbul et al.
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported outcomes.
mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
cell-mediated immune function, ↑ IGF-1 Debats et al.
14 days Surgery 30 17 g Arg
Barbul et al. [59]
Not supplemented
Nussbaum
Not supplemented
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported
↑
14 days Surgery 36 24.8 g Arg
Study Duration Patient Population n Dosage Control Group Outcome Barbul et al. [59]
↑
14 days Surgery 30 17 g Arg
cell-mediated immune func-
Not supplemented
Nussbaum
Not supplemented
Patient Population
↑ Collagen deposition, Wound-breaking strength, ↑ Lymphocyte mitogenesis Nussbaum
14 days 36 24.8 g Arg
Study Duration Patient Population n Dosage Control Group
n Dosage Control Group Outcome
Study Duration
14 days Surgery 17 g Arg
↔ Citrulline, ornithine and NO levels, ↔ Angiogenesis, ↔ Reepithelialisation Sigal et al.
Not supplemented
Isonitrogenous solution
↑ Collagen synthesis, cell-mediated immune func-
↑ Lymphocyte mitogenesis
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported
↑ Wound-breaking strength,
14 days Surgery 36 24.8 g Arg
Study Duration Patient Population n Dosage Control Group Outcome
↔
5 days Surgery 35 30 g intravenous Arg
Not supplemented
↑ Collagen deposition, ↑ Wound-breaking strength, ↑ Lymphocyte mitogenesis
Isonitrogenous solution
↑
↑ Collagen deposition, Wound-breaking strength, ↑ Lymphocyte mitogenesis
30 g intravenous Arg
mented
Not supplemented
Not supplemented
Isonitrogenous solution
Not supplemented
↑ Collagen synthesis, cell-mediated immune func-
tion, Isonitrogenous solution
14 days 30 17 g Arg
14 days Surgery 36
5 days Surgery 30 g intravenous Arg
Duration Patient Population n Dosage Control Group Outcome
Isonitrogenous solution
↔ Lymphocyte proliferation,
Barbul et al. [59] 14 days Surgery 36 24.8 g Arg
Not supple-
[59]
7 days Abdominal surgery 14.7 g intravenous Arg
↔ Citrulline, ornithine and NO levels,
17 g Arg
Isonitrogenous solution
Collagen deposition, Wound-breaking strength,
Not supplemented
↑ Collagen synthesis, cell-mediated immune func-
Isonitrogenous solution Langkamp-
Isonitrogenous solution
Not supplemented
14.7 g intravenous Arg
↑ Collagen synthesis, ↑ T-cell-mediated immune function, ↑ IGF-1
Not supple- ↔ Lymphocyte proliferation, ↔
↔ Reepithelialisation Sigal et al.
30 g intravenous Arg
NO levels,
Lymphocyte mitogenesis Not supple-
Not supplemented
Debats et al.
[64]
Elderly people with pressure ulcers
Not supplemented
pressure ulcers
Nussbaum [64] 14 days Surgery 30 17 g Arg
Isonitrogenous ↔ Lymphocyte proliferation,
↔ NO levels,
↑ Collagen synthesis, T-
Henken et al. 4 weeks
0, 8.5 or 17 g Arg
Abdominal surgery 14.7 g intravenous Arg
- [65]
- [66]
Debats et al.
Isonitrogenous solution
Elderly people with Not supplemented
Isonitrogenous solution
30 g intravenous Arg
Citrulline, ornithine and NO levels, ↔ Sigal et al. Isonitrogenous
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: ni↔ does not increase or decrease.
14.7 g intravenous Arg
Isonitrogenous solution
Isonitrogenous solution
↔ Lymphocyte prolifera-
Citrulline, ornithine and NO levels,
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔
Not supplemented
↔ Lymphocyte proliferation, ↔
30 14.7 g intravenous Arg
Henken et al. [60]
33 0, 8.5 or 17 g Arg
30 g intravenous Arg
Isonitrogenous solution
tion, Langkamp-
Sigal et al.
Isonitrogenous solution
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔
↔ Citrulline, ornithine and ↔ Angiogenesis,
Debats et al. [65] 5 days Surgery 35
pressure ulcers
Elderly people with
Not supplemented
7 days Abdominal surgery 30 14.7 g intravenous Arg
Isonitrogenous
Collagen Deposition (Hydroxyproline Content)0892
Henken et al. 4 weeks
0, 8.5 or 17 g Arg
Angiogenesis,
↔ Lymphocyte prolifera-
Not supplemented
↔ Lymphocyte proliferation,
Collagen Deposition (Hydroxyproline Content)0892
Reepithelialisation Sigal et al. [66] 7 days
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrea
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( arginine aspartate ( = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group ( trols). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
solution
pressure ulcers
Elderly people with pressure ulcers
Not supplemented
Collagen Deposition (Hydroxyproline Content)0892
Lymphocyte proliferation,
14.7 g intravenous Arg
Isonitrogenous solution
Abdominal surgery
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new tetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( arginine aspartate ( nificant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth f tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
[60]
Henken et al. 4 weeks
33 0, 8.5 or 17 g Arg
Langkamp-
Isonitrogenous solution
↔ Lymphocyte proliferation, ↔ NO
30
Elderly people with pressure ulcers
Not supplemented
↔ Lymphocyte proliferation,
7 days Abdominal surgery 30 14.7 g intravenous Arg
NO LangkampHenken et al. [60]
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new tetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↔ does not increase or decrea
4 weeks
33 0, 8.5 or 17 g Arg
Collagen Deposition (Hydroxyproline Content)0892
Lymphocyte proliferation,
Elderly people with pressure ulcers
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth f tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
Not supplemented
Collagen Deposition (Hydroxyproline Content)0892
Elderly people with pressure ulcers
Not supple- ↔ Lymphocyte prolifera↔ NO, ↔ IL-2
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
4 weeks
33 0, 8.5 or 17 g Arg
NO,
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑ ↔ does not increase or decr
33 0, 8.5 or 17 g Arg
Collagen Deposition (Hydroxyproline Content)0892
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
IL-2
Collagen Deposition (Hydroxyproline Content)0892
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases;
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
Collagen Deposition (Hydroxyproline Content)0892
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
does not increase or decrease.
lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Collagen Deposition (Hydroxyproline Content)0892
Collagen Deposition (Hydroxyproline Content)
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune func-
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double-blind controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group,
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double-blind controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group, p < 0.001). Meanwhile, the increment in re-epithelialization failed to reach significance (85 ± 7.1% in the arginine group vs. 81 ± 8.5% in the placebo group, p > 0.05).
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double-blind controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n = 19) for 10 days. Angiogenesis, assessed as the number of vessels per high power field significantly increased on day 10 (8.9 ± 3.1 in the arginine group vs. 8 ± 2.8 in the placebo group, p < 0.001).
In a double-blind RCT conducted and involving 30 adults for 7 days by Sigal et al. [66], the enhancement of lymphocyte proliferation was not observed (p > 0.05) after intravenous supplementation with 14.7 g of arginine compared to controls, treated with Travasol 10%
In a double-blind RCT conducted and involving 30 adults for 7 days by Sigal et al. [66], the enhancement of lymphocyte proliferation was not observed (p > 0.05) after intra-
- Nutrients 2021, 13, 2498 10 of 26
(an isonitrogenous mix of amino acids). The nitrogen balance measured in the supplemented group (−8.8 g/day) was comparable to the control group (−9.2 g/day, p > 0.05).
supplemented group (−8.8 g/day) was comparable to the control group (−9.2 g/day, p >
- 0.05). Langkamp-Henken et al. [60], also conducted an RCT on 33 elderly patients supple-
- 1.25).
Langkamp-Henken et al. [60], also conducted an RCT on 33 elderly patients supplemented with different amounts of arginine (0, 8.5, or 17 g) for 4 weeks. These amounts of arginine used represented approximately 2.2 and 4.5% of an 1800 kcal intake, respectively. The mean daily intake of the patients in this study ranged between 1713 and 2474 kcal with an additional 2.4 and 3.3 g of arginine. After the treatment, no increase in lymphocyte proliferation was observed between groups, while NO increased, although not statistically significant (p > 0.05).
Effects of arginine supplementation on collagen deposition, measured by hydroxyproline content were reported in 2 studies. The study conducted by Barbul et al. [59] reported two results due to two different amounts of supplementation given (17 and 24.8 g arginine). Compared to control, arginine supplementation significantly enhanced hydroxyproline content (MD: 4.49, 95% CI: 3.54, 5.45, p < 0.00001, Figure 8). There was a high heterogeneity (I2 = 99%) among studies. However, there was a lack of significance between the intervention and control group in the Nussbaum study [64] (MD: −0.01, 95% CI: −1.27, 1.25).
Figure 8. Hydroxyproline content: fixed-effects meta-analysis and forest plot from studies providing supplementation of arginine.
3.4. Glutamine (Gln)
3.4. Glutamine (Gln)
Glutamine has been shown to influence several parameters involved in wound healing. This behavior was confirmed by many of the randomized controlled trials that will be reported below, where patients with a total, partial parenteral or early enteral nutrition were supplemented with alanine-glutamine dipeptide, in a concentration ranging from 0.2 to 0.5 g Ala-Gln/kg/day for up to 14 days, as summarized in Table 3. Ala-Gln dipeptide is usually used instead of free Gln, due to its heat stability and its rapid hydrolyzation to free amino acids in plasma [68].
Glutamine has been shown to influence several parameters involved in wound healing. This behavior was confirmed by many of the randomized controlled trials that will be reported below, where patients with a total, partial parenteral or early enteral nutrition were supplemented with alanine-glutamine dipeptide, in a concentration ranging from 0.2 to 0.5 g Ala-Gln/kg/day for up to 14 days, as summarized in Table 3. Ala-Gln dipeptide is usually used instead of free Gln, due to its heat stability and its rapid hydrolyzation to free amino acids in plasma [68].
NO levels, ↔
Citrulline, ornithine and NO levels,
Wound-breaking strength, ↑ Lymphocyte mitogenesis Nussbaum [64]
- Nutrients 2021, 13, 2498 11 of 26
Studies evaluating the effect of arginine supplementation on wound healing and reported
5 days Surgery 35
5 days Surgery 35 30 g intravenous Arg
NO levels, ↔ Angiogenesis,
↔
[65]
[65]
solution
solution
Abdominal surgery LangkampHenken et al. [60]
3.3. Arginine (Arg)
[65]
[66]
Table 3. Studies evaluating the effect of glutamine supplementation on wound healing and reported outcomes.
Sigal et al. [66]
Sigal et al. [66]
Study Duration Patient Population n Dosage
14 days Surgery 30 17 g Arg
7 days 30
7 days Abdominal surgery 30 14.7 g intravenous Arg
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
mented
solution tion, ↔ NO LangkampHenken et al. [60]
solution tion, LangkampHenken et al. [60]
↑ Collagen deposition,
tion, ↑ IGF-1 Debats et al. [65]
Sigal et al. [66]
Isonitrogenous solution
↔
Dosage
7 days Abdominal surgery
4 weeks
33 0, 8.5 or 17 g Arg
14 days Surgery 24.8 g Arg
↔ Citrulline, ornithine and
pressure ulcers
mented
Elderly people with pressure ulcers
Not supple-
↔ Lymphocyte prolifera-
Study Duration Patient Population n
Control Group Outcome Gln (g/kg BW/day)
[59]
mented
Gln Dip (g/kg BW/day Ala-Gln)
Isonitrogenous solution
4 weeks 33
4 weeks
0, 8.5 or 17 g Arg
5 days Surgery 35 30 g intravenous Arg
LangkampHenken et al. [60]
Elderly people with pressure ulcers
Not supplemented
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth f tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
↑ Collagen synthesis, cell-mediated immune func-
4 weeks
33 0, 8.5 or 17 g Arg
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported
↔ Reepithelialisation Sigal et al.
Not supplemented
↑ T-cell lymphocytes,
IL-2,
Isonitrogenous solution
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; : increases; ↔ does not increase or decr
-hydroxy-β- -1: insulin-like growth factor; NO: ni↔
Surgery 17 g Arg
O’riordain et al. [69] 5 days Surgery 22 0.18 0.27
Isonitrogenous solution
↔ Lymphocyte prolifera-
Abdominal surgery 30 14.7 g intravenous Arg
TNFα De Beaux et al. [70] 7 days Critical illness 14 0.22 0.33
IL-6,
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth tric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
tion, ↔ Langkamp-
Dosage Control Group Outcome
Collagen Deposition (Hydroxyproline Content)0892
Isonitrogenous
↔ Citrulline, ornithine and NO levels, Angiogenesis,
solution ↑ Lymphocytes, ↓ IL-8,
IL-6
↑ Collagen deposition, Wound-breaking strength, ↑ Lymphocyte mitogenesis Nussbaum
Collagen Deposition ( )0892
Collagen Deposition (Hydroxyproline Content)0892
Debats et al.
Isonitrogenous solution
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Not supplemented
↔ Lymphocyte proliferation, ↔ NO, ↔
Not supplemented
30 g intravenous Arg
↑ NO, ↓ LOS and patient mortality, ↑ Mood and general well-being Jacobi et al. [72] 7 days Surgery 34 0.27 0.4
0, 8.5 or 17 g Arg
24.8 g Arg
Isonitrogenous solution
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate ( = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm;
pressure ulcers
Collagen Deposition (Hydroxyproline Content)0892
[59]
Morlion et al. [71] 5 days Surgery 28 0.2 0.3
↔ Abdominal surgery 14.7 g intravenous Arg
[60]
-
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. improvement in a randomized, controlled trial (RCT) in 36 healthy and n mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group ( trols). These results were confirmed later by Nussbaum trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the hydroxyproline deposition (17.4 ± 2 nmol/cm; p < 0.02) and T-cell-mediated immune function.
Isonitrogenous solution
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
Collagen synthesis, cell-mediated immune func-
[67]. Barbul et al. [59] observed this on-smoking hu-
Isonitrogenous solution
Not supple-
IL-10, ↑ Wound healing Jiang et al. [73] 7 days Abdominal surgery 60 0.34 0.5
30
Isonitrogenous
tion, Debats et al.
Elderly people with
Not supple-
solution ↑ NO, ↓ LOS Powell-Tuck et al. [74] 4–16.5 days Mixed 168 0.26 0.38
Collagen Deposition (Hydroxyproline Content)0892
Henken et al.
0, 8.5 or 17 g Arg
Isonitrogenous
Isonitrogenous
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group ( < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the
solution ↓ LOS and patient mortality Mertes et al. [75] 6 days Abdominal surgery 37 0.34 0.5
35 30 g intravenous Arg
NO levels, ↔ Angiogenesis, ↔ Reepithelialisation
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
Isonitrogenous
IL-6 Karwowska et al. [76] 10 days Surgery 30 0.2 0.3
solution ↑ NO, ↓ LOS,
p
Isonitrogenous solution
↔ Lymphocyte proliferation, ↔
Abdominal surgery 30 14.7 g intravenous Arg
↑ IgA and IgG, ↑ T-cell lymphocytes, ↓ LOS Neri et al. [77] >7 days Surgery 33 0.2 0.3
Isonitrogenous solution
[66]
Collagen Deposition (Hydroxyproline Content)0892
p
Isonitrogenous
Several studies have demonstrated that supplementation with arginine i lagen deposition and, therefore, enhances wound-breaking strength. The wound ing strength is the force needed to disrupt a wound [67]. Barbul et al. [59] improvement in a randomized, controlled trial (RCT) in 36 healthy and non mans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and
Elderly people with
Not supple- ↔ Lymphocyte prolifera-
[64]
solution ↓ LOS Wischmeyer et al. [78] >7 days Critical illness 31 0.57 0.85
Henken et al. [60]
33 0, 8.5 or 17 g Arg
Isonitrogenous
solution ↓ CRP Goeters et al. [61] >9 days Surgery 144 0.2 0.3
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
< 0.02) and T-cell-mediated immune func-
Isonitrogenous solution
NO, ↓ Patient mortality Lin et al. [79] 6 days Abdominal surgery 48 0.28 0.417
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo ( Angiogenesis, assessed as the number of vessels per high power field significantly in-
↑ NO, ↓ IL-6, ↑ T-cell lymphocytes Ockenga et al. [80] >7 days Critical illness 28 0.2 0.3
Isonitrogenous solution
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were measured after supplementing 30 g of arginine (n = 16) or placebo (n
Collagen Deposition (Hydroxyproline Content)0892
Arginine also influences the nitrogen balance. Nevertheless, this balance improvement has been reported in many, but not in all studies. In a randomized double controlled study by Debats et al. [65], specific parameters related to wound healing were
Isonitrogenous
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-break-
solution ↑ Lymphocytes, ↓ LOS, ↓ CRP
[65]
arginine (n n
tal stay and patient mortality; lactulose/mannitol ratio; C-reactive protein; cytokines (IL-6
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on
- Nutrients 2021, 13, 2498 12 of 26
tal stay and patient mortality; lactulose/mannitol ratio; C-reactive protein; cytokines (IL-
Table 3. Cont.
Table 2. outcomes.
Duration Patient Population
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
were identified under glutamine: nitrogen balance; wound healing time; length of hospital stay and patient mortality; lactulose/mannitol ratio; C-reactive protein; cytokines (IL levels, TNFα levels) and T-cell lymphocytes.
↑ Collagen deposition, Wound-breaking strength,
Dosage
14 days Surgery 36 24.8 g Arg
Study Duration Patient Population n Dosage Control Group Outcome Barbul et al.
Study Duration Patient Population n
Control Group Outcome Gln (g/kg BW/day)
Gln Dip (g/kg BW/day Ala-Gln)
[59]
Lymphocyte mitogenesis
Not supplemented
↑ Collagen synthesis, cell-mediated immune function, IGF-1 Debats et al. [65]
14 days Surgery 36 24.8 g Arg
Wound-breaking strength, ↑ Lymphocyte mitogenesis Not supple-
Studies evaluating the effect of arginine supplementation on wound healing and reported
↑ NO, ↓ Infectious complications, ↑ Lymphocytes CD4 CD8 Xian-Li et al. [62] 14 days Critical illness 69 0.27 0.4
Not supplemented
Isonitrogenous solution
30 17 g Arg
Fuentes-Orozco et al. [81] 10 days Surgery 10 0.27 0.4
[64]
Research on the pharmacological effects of arginine supplementation has been mostly based on its use for acute wounds, although some trials have studied its effect on chronic wounds (Table 2).
Collagen synthesis,
Dosage Control Group
Isonitrogenous
↔ Citrulline, ornithine and NO levels, ↔ Angiogenesis, ↔ Reepithelialisation Sigal et al. [66]
cell-mediated immune function, ↑ IGF-1 Debats et al.
solution ↓ LOS and patient mortality Zhou et al. [82] 12 days Critical illness 30 0.34 0.5
Isonitrogenous solution
↑ Collagen deposition, Wound-breaking strength, ↑ Lymphocyte mitogenesis Nussbaum
30 g intravenous Arg
Not supplemented
Isonitrogenous solution
↑ Wound healing, ↓ Intestinal permeability
36 24.8 g Arg
↔ Citrulline, ornithine and NO levels, ↔ Angiogenesis, ↔ Reepithelialisation
[59]
Table 2. Studies evaluating the effect of arginine supplementation on wound healing and reported outcomes.
Isonitrogenous solution
Isonitrogenous solution
↔ Lymphocyte proliferation, ↔
Quan et al. [83] 7 days Abdominal surgery 20 0.53 0.78 Not specified ↓ Intestinal permeability
30 g intravenous Arg
Abdominal surgery 30 14.7 g intravenous Arg
↑ Collagen synthesis, cell-mediated immune func-
Isonitrogenous
Not supple-
Kłek et al. [84] 12 days Surgery 105 0.27 0.4
solution ↑ Lymphocytes, ↓ LOS Lin et al. [85] 6 days Abdominal surgery 48 0.28 0.417
30
Patient Population Dosage Control Group Barbul et al.
Isonitrogenous
Isonitrogenous
Elderly people with pressure ulcers
Not supple- ↔ Lymphocyte proliferation,
tion, Debats et al.
Abdominal surgery 30 14.7 g intravenous Arg
solution ↑ NO, ↓ IL-6 Yao et al. [86] 5 days Surgery 40 0.34 0.5
Henken et al. [60]
33 0, 8.5 or 17 g Arg
[66]
tion, ↔ Langkamp-
Not supplemented
24.8 g Arg
Isonitrogenous
Isonitrogenous solution
solution ↓ LOS, ↑ CD14 Déchelotte et al. [87] 5 days Surgery 143 0.3 0.45
Elderly people with
Not supplemented
↔ Lymphocyte proliferation, ↔ NO, ↔
35 30 g intravenous Arg
NO levels, ↔
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
0, 8.5 or 17 g Arg
↑ Collagen synthesis,
↔ Reepithelialisation Sigal et al. [66]
Isonitrogenous solution
LOS, ↑ NO, ↓ Intestinal
[60]
Nussbaum
Not supplemented
↔ Lymphocyte proliferation, ↔ NO
Surgery 17 g Arg
permeability S¸ahin et al. [88] 10.5 ± 3.6 days Critical illness 40 0.3 0.45
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
14.7 g intravenous Arg
Isonitrogenous solution
Collagen Deposition (Hydroxyproline Content)0892
solution
T-cell lymphocytes, ↓ LOS Cai et al. [89] 14 days Critical illness 110 0.19 0.29
↔ Citrulline, ornithine and NO levels,
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and
Isonitrogenous
Elderly people with
Not supplemented
Lymphocyte proliferation, ↔ NO,
Debats et al.
Isonitrogenous solution
solution ↑ T-cell lymphocytes, ↓ CRP Duška et al. [90] 13 days Critical illness 30 0.2 0.3
Henken et al. [60]
4 weeks
33 0, 8.5 or 17 g Arg
Surgery 30 g intravenous Arg
Collagen Deposition (Hydroxyproline Content)0892
Isonitrogenous
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride ( = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in con-
solution ↑ NO Estívariz et al. [91] 7 days Surgery 63 0.34 0.5
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
Sigal et al.
Isonitrogenous solution
14.7 g intravenous Arg
Isonitrogenous solution
T-cell Lymphocytes,
↓ Nosocomial infections Dong et al. [92] 6 days Abdominal surgery 40 0.35 0.5
↑ T-cell lymphocytes, ↓ TNFα, ↓ IL-2R Fuentes-Orozco et al. [93] 10 days Critical illness 44 0.27 0.4
Isonitrogenous solution
Elderly people with pressure ulcers
Not supplemented
↔
Collagen Deposition (Hydroxyproline Content)0892
Henken et al.
0, 8.5 or 17 g Arg
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline con-
↑ T-cell lymphocytes, ↑ IgA, ↑ NO, ↓ CRP, ↑ IL-10, ↓ IL-6 Yeh et al. [94] 7 days Surgery 70 0.2 0.29
Isonitrogenous solution
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decr
= 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group ( < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per day for 14 days. An improvement was also observed in collagen synthesis through the
Isonitrogenous solution
↓ CRP, ↓ LOS,
Patient mortality
5 days preoperatively
Isonitrogenous
Collagen Deposition (Hydroxyproline Content)0892 Several studies have demonstrated that supplementation with arginine i
Asprer et al. [95]
Abdominal surgery 34 0.2 0.3
solution ↑ Lymphocytes
Citrulline, ornithine and NO levels, ↔ Angiogenesis,
Lymphocyte mitogenesis Nussbaum Not supple-
- Nutrients 2021, 13, 2498 13 of 26
5 days Surgery 35 30 g intravenous Arg
Collagen synthesis, T
3.3. Arginine (Arg)
[65]
solution
mostly based on its use for acute wounds, although some trials have studied its effect on
7 days Abdominal surgery Langkamp-
Table 3. Cont.
Debats et al.
Isonitrogenous
5 days Surgery 35 30 g intravenous Arg
NO levels, ↔ Angiogenesis, ↔ Reepithelialisation
Dosage
Elderly people with pressure ulcers
Henken et al. 4 weeks
33 0, 8.5 or 17 g Arg
Studies evaluating the effect of arginine supplementation on wound healing and reported
Study Duration Patient Population n
Control Group Outcome Gln (g/kg BW/day)
Gln Dip (g/kg BW/day Ala-Gln) Engel et al. [63] 3 days Surgery 58 0.5 0.74
tion,
Isonitrogenous
7 days Abdominal surgery 30 14.7 g intravenous Arg
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
solution tion, ↔ NO LangkampHenken et al. [60]
[66]
Patient Population n Dosage Control Group
Isonitrogenous solution
T-cell lymphocytes Fan et al. [96] 7 days Abdominal surgery 40 0.13 0.2
Collagen deposition, ↑ Wound-breaking strength, ↑ Lymphocyte mitogenesis
Not supplemented
↔ Lymphocyte proliferation, ↔ NO,
↓ LOS and infectious complications Quan et al. [97] 4 days Abdominal surgery 20 0.35 0.5 Normal saline ↑ NO, ↓ IL-6
Isonitrogenous solution
4 weeks 33 0, 8.5 or 17 g Arg
Surgery 36 24.8 g Arg
Collagen Deposition (Hydroxyproline Content)0892
mented
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and arginine aspartate (p = 0.028, 17.57 ± 2.16 nmol/cm) groups, respectively, following a significant difference observed in the controlled group (p < 0.001; 10.1 ± 2.32 nmol/cm in controls). These results were confirmed later by Nussbaum [64], who carried out a similar trial in 45 healthy elderly people, randomly supplemented or not with 17 g of arginine per
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔ does not increase or decrease.
↑ cell-mediated immune func-
Isonitrogenous solution
Not supple-
Andrews et al. [98] Up to 7 days Mixed 502 0.2 0.3
Patient mortality Çekmen et al. [99] >5 days Mixed 30 0.35 0.5
Surgery 30 17 g Arg
↓ CRP, ↓ LOS and patient mortality Grau et al. [100] 5–9 days Mixed 127 0.35 0.5
Isonitrogenous solution
tion,
Collagen Deposition (Hydroxyproline Content)0892
Isonitrogenous
Isonitrogenous
Several studies have demonstrated that supplementation with arginine increases collagen deposition and, therefore, enhances wound-breaking strength. The wound-breaking strength is the force needed to disrupt a wound [67]. Barbul et al. [59] observed this improvement in a randomized, controlled trial (RCT) in 36 healthy and non-smoking humans by supplementing their diet with 24.8 g of free arginine as arginine hydrochloride and 17 g of free arginine as arginine aspartate per day for 2 weeks. Hydroxyproline content was assessed as an index of the synthesis and deposition of new collagen in a polytetrafluoroethylene tube inserted in the wound site. An enhanced collagen deposition at 137 and 74% was noted in the arginine hydrochloride (p = 0.028; 23.85 ± 2.16 nmol/cm) and
solution ↓ Nosocomial infections Lu et al. [101] 7 days Surgery 50 0.3 0.45
Surgery 35 30 g intravenous Arg
NO levels, ↔ Angiogenesis,
Isonitrogenous solution
↑ NO, ↓ IL-6, ↓ CRP, ↓ Infectious complications
Reepithelialisation Abdominal surgery 30 14.7 g intravenous Arg
Isonitrogenous
Wernerman et al. [102] 7 days Mixed 413 0.28 0.42 Normal saline ↓ LOS and patient mortality
solution tion, Elderly people with pressure ulcers
Isonitrogenous
solution ↓ TNFα Richard et al. [104]
Xu et al. [103] 12 days Critical illness 80 Unknown Unknown
3 days pre, 4 days postoperatively
Surgery 22 0.53 0.78 Not supplemented ↓ LOS and patient mortality, ↓
33 0, 8.5 or 17 g Arg
mented ↔
CRP
Ala: alanine; BW: body weight; CRP: C-reactive protein; Gln: glutamine; Gln dip: glutamine dipeptide; IgA: immunoglobulin A; IL-2R: interleukin-2 receptor; IL-6: interleukin-6; LOS: length of stay; NO: nitric oxide; TNFα: tumor necrosis factor α; ↑: increases; ↓: decreases;
Arg: arginine; Cu: copper; Gln: glutamine; HBM: β-hydroxy-β-methylbutyrate; IGF-1: insulin-like growth factor; NO: nitric oxide; P: phosphorous; RME: resting metabolic expenditure; Zn: zinc; ↑: increases; ↔does not increase or decrease.does not increase or decrease.
3.4.1. Nitrogen Balance
3.4.1. Nitrogen Balance
In two double-blind RCT performed in 48 patients supplemented with 0.28 g/kg/day Gln for 6 days, by Lin et al. [79,85], higher nitrogen balance, although not significant (−3.2 ± 1.6 vs. −6.5 ± 2.7 g N, p > 0.05) was observed after Gln treatment. Cumulative nitrogen balance was higher in Glu-supplemented patients with lower illness severity, assessed with APACHE II scores (acute physiology score + age points + chronic health points), but this was not observed when severity was higher (APACHE II score > 6). However, when compared with controls, the reducing effect was not reported, showing that the improving effect on cumulative nitrogen balance is not due to the reduction of muscle protein breakdown, but an enhanced protein synthesis.
In two double-blind RCT performed in 48 patients supplemented with 0.28 g/kg/day Gln for 6 days, by Lin et al. [79,85], higher nitrogen balance, although not significant (−3.2 ± 1.6 vs. −6.5 ± 2.7 g N, p > 0.05) was observed after Gln treatment. Cumulative nitrogen balance was higher in Glu-supplemented patients with lower illness severity, assessed with APACHE II scores (acute physiology score + age points + chronic health points), but this was not observed when severity was higher (APACHE II score > 6). However, when compared with controls, the reducing effect was not reported, showing that the improving effect on cumulative nitrogen balance is not due to the reduction of muscle protein breakdown, but an enhanced protein synthesis.
In contrast, a higher cumulative nitrogen balance, adjusted to standard body surface area, was not noted (−193 ± 50 vs. −198 ± 77 g N, p > 0.05) in a prospective doubleblind RCT conducted by Duška et al. [90], who supplemented 30 patients suffering from critical illnesses with 0.2 g/kg/day Gln for 13 days, and compared the results against the placebo group, treated with isonitrogenous nutrition. Similar results were reported by Mertes et al. [75] in a double-blind RCT on 37 patients undergoing major abdominal surgery for 5 days (−14.1 ± 9.1 vs. −31.7 ± 11.4 g N, p < 0.05). Jiang et al. [73] also noted a rise of the cumulative nitrogen balance (144.3 ± 145.6 vs. −5.1 ± 162.7 mg/kg, p = 0.0004) in a double-blind RCT in 60 patients undergoing abdominal surgery, supplemented with 0.34 g/kg/day or an isonitrogenous solution for 7 days.
In contrast, a higher cumulative nitrogen balance, adjusted to standard body surface area, was not noted (−193 ± 50 vs. −198 ± 77 g N, p > 0.05) in a prospective double-blind RCT conducted by Duška et al. [90], who supplemented 30 patients suffering from critical illnesses with 0.2 g/kg/day Gln for 13 days, and compared the results against the placebo group, treated with isonitrogenous nutrition. Similar results were reported by Mertes et al. [75] in a double-blind RCT on 37 patients undergoing major abdominal surgery for 5 days (−14.1 ± 9.1 vs. −31.7 ± 11.4 g N, p < 0.05). Jiang et al. [73] also noted a rise of the cumulative nitrogen balance (144.3 ± 145.6 vs. −5.1 ± 162.7 mg/kg, p = 0.0004) in a doubleblind RCT in 60 patients undergoing abdominal surgery, supplemented with 0.34 g/kg/day or an isonitrogenous solution for 7 days.
On the contrary, in a double-blind RCT performed by Morlion et al. [71] in 28 patients undergoing surgery, supplemented with 0.2 g/kg/day Gln for 5 days, the increase in cumulative nitrogen balance was also observed (−7.9 ± 3.6 vs. −233.0 ± 2.6, p < 0.01).
On the contrary, in a double-blind RCT performed by Morlion et al. [71] in 28 patients undergoing surgery, supplemented with 0.2 g/kg/day Gln for 5 days, the increase in cumulative nitrogen balance was also observed (−7.9 ± 3.6 vs. −233.0 ± 2.6, p < 0.01).
Eleven studies reported data on nitrogen balance. Compared to control, glutamine supplementation significantly increased nitrogen balance (MD: 0.39, 95% CI: 0.21, 0.58, p < 0.0001, Figure 9). There was a high heterogeneity (I2 = 91%) among studies. Nevertheless, results reported by 3 studies lacked from significance. These studies are Déchelotte et al. [87] (MD: 0.00, 95% CI: −0.37, 0.37), S¸ahin et al. [88] (MD: 0.29, 95% CI: −0.33, 0.92), and Yeh et al. [94] (MD: 0.32, 95% CI: −0.15, 0.79).
Eleven studies reported data on nitrogen balance. Compared to control, glutamine supplementation significantly increased nitrogen balance (MD: 0.39, 95% CI: 0.21, 0.58, < 0.0001, Figure 9). There was a high heterogeneity (I2= 91%) among studies. Nevertheless, results reported by 3 studies lacked from significance. These studies are Déchelotte et al. [87] (MD: 0.00, 95% CI: −0.37, 0.37), Şahin et al. [88] (MD: 0.29, 95% CI: −0.33, 0.92), and Yeh et al. [94] (MD: 0.32, 95% CI: −0.15, 0.79).
Figure 9. Nitrogen balance: fixed-effects meta-analysis and forest plot from studies providing supplementation of gluta-
- 3.4.2. Wound Healing Time
3.4.2. Wound Healing Time
Two trials [72,82] measured the effect of glutamine supplementation on wound healing. Zhou et al. [82] performed a double-blind RCT and noticed a lower infection rate (13 vs. 26%), although this difference failed to reach significance (p > 0.05). Also, it was noted a significant reduction of the wound healing time in the study group (32.1 ± 3.3 days) compared to the control group (36.6 ± 6.6 days, p < 0.012) after supplementing 30 patients suffering from severe burns with 0.34 g/kg/day Gln or an isonitrogenous solution, for 12 days. This data is in agreement with the RCT performed by Jacobi et al. [72] where, after supplementing 34 patients with 0.27 g/kg/day Gln for 7 days, observed a signifi-
Two trials [72,82] measured the effect of glutamine supplementation on wound healing. Zhou et al. [82] performed a double-blind RCT and noticed a lower infection rate (13 vs. 26%), although this difference failed to reach significance (p > 0.05). Also, it was noted a significant reduction of the wound healing time in the study group (32.1 ± 3.3 days) compared to the control group (36.6 ± 6.6 days, p < 0.012) after supplementing 30 patients suffering from severe burns with 0.34 g/kg/day Gln or an isonitrogenous solution, for 12
cant increase in wound healing (p < 0.05), explained in part by the incidence of wound complications post-surgery.
- 3.4.3. Length of Hospital Stay (LOS) and Patient mortality
The decrease in LOS was reported by many studies. Neri et al. [77] studied the effect of 0.2 g/kg/day Gln in 33 patients supplemented for at least 7 days. In this trial, the study group spent 11.5 ± 2 days hospitalized, versus the control group, which spent 15 ± 3 days (p < 0.05). Meanwhile, in the prospective double-blind RTC carried out by Yao et al. [86] in 40 people for 5 days, the study group spent 10.6 ± 1.2 days in the hospital, while the non-supplemented group spent 11.7 ± 2 days (p = 0.03).
These data are in agreement with Fan et al. [96], who performed an RCT in 40 patients undergoing surgery and observed a trend to a reduction both in LOS (22.3 ± 2.1 vs. 24.9 ± 1.7 days, p = 0.32) and infectious complications after supplementation with 0.13 g/kg/day Gln, although these data failed to reach significance. The reduction in LOS and mortality was also observed by Wischmeyer et al. [78] in a double-blind RCT in 31 patients with severe burns, supplemented with 0.57 g/kg/day L-Gln for at least 7 days via parenteral feeding. However, in this case, there was no significant difference seen in the LOS (31 ± 10.1 vs. 30 ± 9.3, p > 0.05) nor mortality (1 vs. 4 deaths, p = 0.19).
Goeters et al. [61] also noticed a patient mortality decline within 6 months (11/33 vs. 21/35 deaths, p < 0.05), resulting in a 66.7% increase of survival of patients treated for ≥ 9 days, compared to 40% in controls, in an unblinded parallel multicentre RCT in 144 critically ill patients supplemented with 0.2 g/kg/day Gln.
Despite the lack of statistical significance, Çekmen et al. [99] also observed a trend to the reduction in LOS (19.3 ± 12.7 vs. 27.5 ± 12.1 days, p = 0.36) and patient mortality (20 vs. 40%, p = 0.42) in a double-blind RCT in 30 critically ill patients supplemented with 0.35 g/kg/day Gln.
Karwowska et al. [76] studied the effect of glutamine supplementation and observed a decrease in LOS; from 15.1 ± 3 days to 12.5 ± 1.2 days (p = 0.005) in the supplemented group. The reduction in LOS was also seen in a double-blind RCT carried out by Powell-Tuck et al. [74] in 168 patients, supplemented with 20 g Gln as part of the nitrogen source of parenteral nutrition or standard feeds, for 7 days. The study group spent an average of 15 fewer days in hospital, compared with the control group (46 ± 10.7 vs. 30 ± 7.2 days, p < 0.03). The latter study failed to demonstrate a decrease in morbidity and mortality in the study group, probably due to the younger age of the patients (48 ± 17 years old vs. 80 ± 19 years old). Nevertheless, this limitation was avoided in a study by Xian-Li et al. [62] in a non-blinded RCT in 69 patients suffering from critical illnesses for 14 days, and it was seen, not only a reduction in LOS (25.3 ± 7.6 vs. 39.1 ± 10.6 days, p < 0.01), but also in patient mortality (0 vs. 43.5%, p < 0.01) after supplementing 0.2 g/kg/day Gln. The same results were obtained by Wernerman et al. [102] in a double-blind RCT, observing a reduction of patient mortality in ICU (8/11 vs. 14/20, p < 0.05) after supplementing 413 patients, in ICU, with 0.28 g/kg/day Gln for 7 days.
On the other hand, Ockenga et al. [80] reported a significant reduction in LOS in a double-blind RCT in 28 patients suffering from acute pancreatitis supplemented with 0.2 g/kg/day Gln for at least 7 days. In this trial, the LOS was an average of 4 days shorter in the study group (21 days) compared to the control group (25 days, p < 0.05) despite the small sample studied. In the trial conducted by Mertes et al. [75] a decrease in hospital stay was also noted (12.8 ± 2.6 vs. 17.5 ± 6.4 days, p < 0.05) compared to the control group. Similar results were observed by Jiang et al. [73] who noted a decrease in 4 days of LOS (12.5 ± 5.1 vs. 16.4 ± 7.1 days, p = 0.02). Morlion et al. [71] not only noted the reduction in LOS (15.5 ± 0.7 vs. 21.7± 2.8 days, p < 0.05), but there were also evident improvements both in mood and general well-being in the supplemented patients.
On the contrary, in a prospective double-blind RCT performed by Déchelotte et al. [87] in 114 patients with critical illnesses treated with 0.33 g/kg/day Gln, no significant differences in LOS nor patient mortality were observed (1.9 vs. 3.8% deaths, p > 0.05), although
On the contrary, in a prospective double-blind RCT performed by Déchelotte et al. [87] in 114 patients with critical illnesses treated with 0.33 g/kg/day Gln, no significant
- Nutrients 2021, 13, 2498 16 of 26
a reduction of the incidence of infectious complications (39 vs. 64%, p < 0.02) was noted. The decrease of nosocomial infections, such as urinary tract infections (2.3 vs. 16.9%, p = 0.03) and pneumonia (8 vs. 29%, p = 0.02) were also observed by Grau et al. [100]. This trial studied the effect of 0.35 g/kg/day Gln in a multicentre, prospective, double-blind, RCT in 502 patients admitted to the UCI. In another double-blind RCT performed by Estívariz et al. [91] in 63 people undergoing surgery and in ICU, patients were supplemented with 0.34 g/kg/day Gln and a lessening of nosocomial infections was observed (13 vs. 36 cases, p < 0.03). Kłek et al. [84] also reported a decrease in postoperative complications (23.3 vs. 36.6%, p < 0.05) and LOS (14.8 vs. 16.4 days, p < 0.05) in an RCT performed in 69 patients undergoing surgery supplemented with 0.27 g/kg/day Gln for 12 days.
noted. The decrease of nosocomial infections, such as urinary tract infections (2.3 vs. 16.9%, p = 0.03) and pneumonia (8 vs. 29%, p = 0.02) were also observed by Grau et al. [100]. This trial studied the effect of 0.35 g/kg/day Gln in a multicentre, prospective, double-blind, RCT in 502 patients admitted to the UCI. In another double-blind RCT performed by Estívariz et al. [91] in 63 people undergoing surgery and in ICU, patients were supplemented with 0.34 g/kg/day Gln and a lessening of nosocomial infections was observed (13 vs. 36 cases, p < 0.03). Kłek et al. [84] also reported a decrease in postoperative complications (23.3 vs. 36.6%, p < 0.05) and LOS (14.8 vs. 16.4 days, p < 0.05) in an RCT performed in 69 patients undergoing surgery supplemented with 0.27 g/kg/day Gln for 12 days.
On the contrary, a double-blind RCT conducted by Andrews et al. [98], patients supplemented with 0.2 g/kg/day Gln for at least 5 days, neither showed a significant improvement in the incidence of infections nor patient mortality compared to the control group (p > 0.05).
On the contrary, a double-blind RCT conducted by Andrews et al. [98], patients supplemented with 0.2 g/kg/day Gln for at least 5 days, neither showed a significant improvement in the incidence of infections nor patient mortality compared to the control group (p > 0.05).
The effect of glutamine on LOS was reported by 21 studies involving 1042 participants. According to the meta-analysis, glutamine supplementation has shown to significantly decrease the days of hospitalization (MD: −2.65, 95% CI: −3.10, −2.21, p < 0.00001, Figure 10). Exhibiting a high heterogeneity among studies (I2 = 84%). Nevertheless, not all the studies showed a significant effect on this parameter. This is the case of the following studies: Wischmeyer et al. [78] (MD: 1.00, 95% CI: −6.51, 8.51), Ockenga et al. [80] (MD: 0.50, 95% CI: −5.00, 6.00), Zhou et al. [82] (MD: −4.00, 95% CI: −8.87, 0.87), Fuentes-Orozco et al. [81] (MD: −0.17, 95% CI: −5.63, 5.29), Kiek et al. [84] (MD: −1.80, 95% CI: −5.68, 2.08), S¸ahin et al. [88] (MD: −2.20, 95% CI: −4.78, 0.38), Fuentes-Orozco et al. [93] (MD: 3.59, 95% CI: −3.47, 10.65), Engel et al. [63] (MD: 0.40, 95% CI: −0.55, 1.35), Çekmen et al. [99] (MD: −8.20, 95% CI: −17.04, 0.64).
The effect of glutamine on LOS was reported by 21 studies involving 1042 participants. According to the meta-analysis, glutamine supplementation has shown to significantly decrease the days of hospitalization (MD: −2.65, 95% CI: −3.10, −2.21, p < 0.00001, Figure 10). Exhibiting a high heterogeneity among studies (I2 = 84%). Nevertheless, not all the studies showed a significant effect on this parameter. This is the case of the following studies: Wischmeyer et al. [78] (MD: 1.00, 95% CI: −6.51, 8.51), Ockenga et al. [80] (MD: 0.50, 95% CI: −5.00, 6.00), Zhou et al. [82] (MD: −4.00, 95% CI: −8.87, 0.87), Fuentes-Orozco et al. [81] (MD: −0.17, 95% CI: −5.63, 5.29), Kiek et al. [84] (MD: −1.80, 95% CI: −5.68, 2.08), Şahin et al. [88] (MD: −2.20, 95% CI: −4.78, 0.38), Fuentes-Orozco et al. [93] (MD: 3.59, 95% CI: −3.47, 10.65), Engel et al. [63] (MD: 0.40, 95% CI: −0.55, 1.35), Ç ekmen et al. [99] (MD: −8.20, 95% CI: −17.04, 0.64).
Figure 10. Length of hospital stay: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
Overall data from 11 studies involving 696 participants (340 and 356 in the study and control group respectively) were evaluated. Patient mortality occurred less frequently in the glutamine-supplemented group than in controls (46 (13.53%) vs. (89 (25%) participants). The odds ratio was 0.48 (95% CI: 0.32, 0.72, p = 0.0004, Figure 11), and heterogeneity among
- Nutrients 2021, 13, 2498 17 of 26
p
studies was 14%. The number needed to treat was 10. In other words, 10 patients would need to receive glutamine supplementation to prevent an additional fatality.
- Figure 11. Patient mortality: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
- Figure 12. Lactulose/mannitol ratio: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
- -
3.4
p
p
p
Five studies involving 210 participants reported data on intestinal permeability, as-
−0 −0 −0 p 2 was 97%.
- of glutamine.
3.4.5. C-Reactive Protein
3.4 -
Çekmen et al. [99] observed a trend to the reduction in CRP (57.65 ± 41.81 vs. 82.87 ± 69.41 mg/L, p = 0.32) compared to the control group. Nevertheless, these results did not reach significance.
p
A double-blind RCT performed by Lu et al. [101] on 50 patients, suffering from gastrointestinal cancer and undergoing surgery, supplemented with 0.3 g/kg/day Gln; reported a significant reduction in CRP serum level (16.7 ± 11.8 vs. 35.2 ± 24.8 mg/dL, p = 0.013). Besides, in the control group, there were noted 4 cases of infectious complications while none were observed in the study group (p = 0.037). The reduction in CRP (p < 0.01)
surgery, supplemented with 0.3 g/kg/day Gln; reported a significant reduction in CRP serum level (16.7 ± 11.8 vs. 35.2 ± 24.8 mg/dL, p
- Nutrients 2021, 13, 2498 18 of 26
was also observed by Wischmeyer et al. [78] in a double-blind RCT in 31 patients with severe burns, supplemented with 0.57 g/kg/day L-Gln for at least 7 days via parenteral feeding. Ockenga et al. [80], as well as Dong et al. [92] also reported a significant decrease in CRP (30 ± 42 vs. 34 ± 51, p < 0.01), (69 ± 19 vs. 99 ± 44, p = 0.01)
was also observed by Wischmeyer et al. [78] in a double-blind RCT in 31 patients with severe burns, supplemented with 0.57 g/kg/day L-Gln for at least 7 days via parenteral feeding. Ockenga et al. [80], as well as Dong et al. [92] also reported a significant decrease in CRP (30 ± 42 vs. 34 ± 51, p < 0.01), (69 ± 19 vs. 99 ± 44, p = 0.01)
Yeh et al. [94] noted the CRP lessening (p = 0.012) in an RCT in 70 patients undergoing surgery for 7 days. Another RCT performed by Cai et al. [89] suggested a pronounced decrease of CRP (25.8 ± 4.9 vs. 15.6 ± 4.2 mg/dL, p = 0.001) after performing an RCT supplementing 110 patients suffering from a critical illness with 0.19 g/kg/day Gln for 14 days. Besides, it was also suggested a decrease in the illness severity, assessed by APACHE II scores (10.35 ± 4.35 vs. 17.75 ± 4.46, p = 0.001). Richard et al. [104] supplemented patients before and after surgery and also observed a significant decrease in CRP (44 ± 35 vs. 69 ± 19 mg/L, p = 0.028)
Yeh et al. [94] noted the CRP lessening (p = 0.012) in an RCT in 70 patients undergoing surgery for 7 days. Another RCT performed by Cai et al. [89] suggested a pronounced decrease of CRP (25.8 ± 4.9 vs. 15.6 ± 4.2 mg/dL, p = 0.001) after performing an RCT supplementing 110 patients suffering from a critical illness with 0.19 g/kg/day Gln for 14 days. Besides, it was also suggested a decrease in the illness severity, assessed by APACHE II scores (10.35 ± 4.35 vs. 17.75 ± 4.46, p = 0.001). Richard et al. [104] supplemented patients before and after surgery and also observed a significant decrease in CRP (44 ± 35 vs. 69 ± 19 mg/L, p = 0.028)
Eleven studies reported the effect of glutamine supplementation on CRP levels. Although, individually, 5 studies data did not reach statistical significance [63,80,87,88,99], the overall effect showed the reduction of CRP levels after supplementation (MD: −1.10, 95% CI: −1.26, −0.93, p < 0.00001, Figure 13). The heterogeneity among studies was 82%.
Eleven studies reported the effect of glutamine supplementation on CRP levels. Although, individually, 5 studies data did not reach statistical significance [63,80,87,88,99], the overall effect showed the reduction of CRP levels after supplementation (MD: −1.10, 95% CI: −1.26, −0.93, p < 0.00001, Figure 13). The heterogeneity among studies was 82%.
Figure 13. C-reactive protein: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
3.4.6. Cytokines
3.4.6. Cytokines
Quan et al. [97] observed a significant reduction of IL-6 (6.95 ± 5.08 vs. 12.88 ± 3.85 ng/L, p < 0.05) when 20 patients undergoing abdominal surgery were supplemented with 0.28 g/kg/day Gln for 4 days in a multicentre double-blind RCT. The reduction in IL6 was also reported by Lu et al. [101] (19.2 ± 9.8 vs. 34.7 ± 18.7 pg/mL, p = 0.01).
Quan et al. [97] observed a significant reduction of IL-6 (6.95 ± 5.08 vs. 12.88 ± 3.85 ng/L, p < 0.05) when 20 patients undergoing abdominal surgery were supplemented with 0.28 g/kg/day Gln for 4 days in a multicentre double-blind RCT. The reduction in IL-6 was also reported by Lu et al. [101] (19.2 ± 9.8 vs. 34.7 ± 18.7 pg/mL, p = 0.01).
On the other hand, Xu et al. [103] noted a reduction in TNFα levels (p = 0.01) in an RCT in 80 patients supplemented with glutamine administered via early enteral nutrition.
On the other hand, Xu et al. [103] noted a reduction in TNFα levels (p = 0.01) in an RCT in 80 patients supplemented with glutamine administered via early enteral nutrition.
O’Riordain et al. [69] performed a double-blind RCT in 22 patients undergoing surgery and supplemented with 0.18 g/kg/day Gln in the form of glycyl-glutamine, for 5 days. This trial measured the IL-6 and TNFα levels. Nevertheless, the data failed to reach significance (p = 0.27 and p > 0.05, respectively). The lack of significance regarding the production of IL-6 was also observed in a double-blind RCT performed by De Beaux et al. [70] in 14 patients suffering from critical illnesses.
O’Riordain et al. [69] performed a double-blind RCT in 22 patients undergoing surgery and supplemented with 0.18 g/kg/day Gln in the form of glycyl-glutamine, for 5 days. This trial measured the IL-6 and TNFα levels. Nevertheless, the data failed to reach significance (p = 0.27 and p > 0.05, respectively). The lack of significance regarding the production of IL-6 was also observed in a double-blind RCT performed by De Beaux et al. [70] in 14 patients suffering from critical illnesses.
Five and four studies reported data on IL-6 and TNFα levels, respectively. Both parameters showed a significant reduction after glutamine supplementation for IL-6 levels (MD: −5.78, 95% CI: −8.71, −2.86, I2 = 74, p = 0.0001) and for TNFα levels (MD: −8.15, 95% CI: −9.34, −6.96, I2 = 97, p < 0.00001, Figure 14a,b). This occurred despite the lack of significance shown by 2 different studies in both parameters. For IL-6 levels this was the case of Mertes et al. [75] (MD: 24.00, 95% CI: −78.69, 126.69) and Lin et al. [85] (MD: −4.5, 95% CI: −9.96, 0.96) while for TNFα levels they were Mertes et al. [75] (MD: 7.00, 95% CI: −0.69, 14.69) and Lu et al. [101] MD: −2.60, 95% CI: −9.78, 4.58).
Five and four studies reported data on IL-6 and TNFα levels, respectively. Both parameters showed a significant reduction after glutamine supplementation for IL-6 levels (MD: −5.78, 95% CI: −8.71, −2.86, I = 74, p = 0.0001) and for TNFα levels (MD: −8.15, 95%
Nutrients 2021, 13, 2498 Mertes et al. [75] (MD: 24.00, 95% CI: −78.69, 126.69) and Lin et al. [85] (MD: −4.5, 95% CI:19 of 26 −9.96, 0.96) while for TNFα levels they were Mertes et al. [75] (MD: 7.00, 95% CI: −0.69,
- (a)
- (b)
Figure 14. (a) IL-6 levels: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine (b) TNFα levels: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
Figure 14. (a) IL-6 levels: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine (b) TNFα levels: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
3.4.7. T-Cell Lymphocytes
3.4.7. T-Cell Lymphocytes
Karwowska et al. [76] performed an RTC and studied the effect of glutamine in the immune system by the increase of T-cells, particularly, CD4 and CD8 lymphocytes, IgA, IgG in well-nourished male patients undergoing abdominal surgery, supplemented with 0.202 g/kg/day Gln. Significant increases in the total counts of lymphocytes (p = 0.005), CD4 lymphocytes (p = 0.005), CD8 lymphocytes (p = 0.01), IgA (p = 0.005) and IgG (p = 0.02) were observed. Kłek et al. [84] also reported an increase in T-cell lymphocytes in the group supplemented with Gln. Nevertheless, this measurement did not reach significance (p > 0.05).
Karwowska et al. [76] performed an RTC and studied the effect of glutamine in the immune system by the increase of T-cells, particularly, CD4 and CD8 lymphocytes, IgA, IgG in well-nourished male patients undergoing abdominal surgery, supplemented with 0.202 g/kg/day Gln. Significant increases in the total counts of lymphocytes (p = 0.005), CD4 lymphocytes (p = 0.005), CD8 lymphocytes (p = 0.01), IgA (p = 0.005) and IgG (p = 0.02) were observed. Kłek et al. [84] also reported an increase in T-cell lymphocytes in the group supplemented with Gln. Nevertheless, this measurement did not reach significance (p 0.05).
Asprer et al. [95] also noticed an increase in T-cell lymphocytes count (p = 0.049) in a prospective RCT performed in 34 patients supplemented with 0.3 g/kg/day Gln for 5 days before surgery. This data is in agreement with Ockenga et al. [80], who also reported an increase in lymphocytes (1.7 ± 0.7 vs. 1.5 ± 0.4, p < 0.01). Similar results were reported by Cai et al. [89] (1.91 ± 0.33 vs. 1.35 ± 0.25 × 109/L, p = 0.001) and Dong et al. [92] (p < 0.01).
Asprer et al. [95] also noticed an increase in T-cell lymphocytes count (p = 0.049) in a prospective RCT performed in 34 patients supplemented with 0.3 g/kg/day Gln for 5 days before surgery. This data is in agreement with Ockenga et al. [80], who also reported an increase in lymphocytes (1.7 ± 0.7 vs. 1.5 ± 0.4, p < 0.01). Similar results were reported by Cai et al. [89] (1.91 ± 0.33 vs. 1.35 ± 0.25 × 109/L, p = 0.001) and Dong et al. [92] (p < 0.01).
On the contrary, O’Riordain et al. [69] reported a significant increase in the synthesis of T-cell DNA, measured with the tritiated thymidine index, compared to the control group (p < 0.05).
On the contrary, O’Riordain et al. [69] reported a significant increase in the synthesis of T-cell DNA, measured with the tritiated thymidine index, compared to the control group (p
In a double-blind RCT carried out by Fuentes-Orozco et al. [81] for 10 days, on 33 patients with secondary peritonitis, supplemented with 0.26 g/kg/day Gln, it was also suggested an increase in T-cells levels, including, their subpopulations, CD4 and CD8. However, no significant difference was reached (p > 0.05).
In a double-blind RCT carried out by Fuentes-Orozco et al. [81] for 10 days, on 33 patients with secondary peritonitis, supplemented with 0.26 g/kg/day Gln, it was also suggested an increase in T-cells levels, including, their subpopulations, CD4 and CD8. However, no significant difference was reached (p > 0.05).
Regarding the effect of glutamine supplementation on T-cell lymphocytes, there is contradictory evidence in the literature.
Regarding the effect of glutamine supplementation on T-cell lymphocytes, there is contradictory evidence in the literature.
However, fourteen studies involving 540 participants (315 and 312 in the experimental and control group, respectively) showed that glutamine supplementation might increase T-cell lymphocytes levels. Nevertheless, the data analysis showed a lack of significance in the measured parameter (MD: −0.16, 95% CI: −0.33, 0.01, I2 = 93, p = 0.07, Figure 15).
Nutrients 2021, 13, 2498 increase T-cell lymphocytes levels. Nevertheless, the data analysis showed a lack of sig-20 of 26 nificance in the measured parameter (MD: −0.16, 95% CI: −0.33, 0.01, I2= 93, p = 0.07, Figure
Figure 15. T-cell lymphocytes: fixed-effects meta-analysis and forest plot from studies providing supplementation of glutamine.
4. Discussion
4. Discussion
- 4.1. Arginine
- 4.2. Glutamine
- 4.1. Arginine
- 4.2. Glutamine
Regarding glutamine supplementation, the greater cumulative nitrogen balance noted in some of these studies explains the use of glutamine by the body as a substrate for the synthesis of NO. Improved nitrogen retention is associated with a shorter length of hospital stay, and high levels of IL-6 are associated with infections and mortality. The lower production of proinflammatory cytokines might also be explained due to the decrease in intestinal permeability [48]. Therefore, an increase in the nitrogen balance and decline of
Regarding glutamine supplementation, the greater cumulative nitrogen balance noted in some of these studies explains the use of glutamine by the body as a substrate for the synthesis of NO. Improved nitrogen retention is associated with a shorter length of hospital stay, and high levels of IL-6 are associated with infections and mortality. The lower production of proinflammatory cytokines might also be explained due to the decrease in intestinal permeability [48]. Therefore, an increase in the nitrogen balance and decline of IL-6 may explain why in many studies, there was a reduction in the length of
- IL-6 may explain why in many studies, there was a reduction in the length of hospitalization and patient mortality observed [61]. However, the decrease in mortality was not always significant in this review, hence not fully agreeing with the review conducted by Bollhalder et al. [53].
In a prospective double-blind RCT performed by Déchelotte et al. [87] in 114 patients with critical illnesses, no significant differences in LOS nor patient mortality were observed (1.9 vs. 3.8% deaths, p > 0.05), and even if there was an improvement in the nitrogen
balance, this variation did not reach significance (−2.44 ± 8.6 vs. −4.4 ± 13.2, p > 0.05). In this case, the benefits of supplementation may have been due to the decrease in intestinal permeability [40,44,46], resulting thus, in the reduction of the incidence of infectious complications (39 vs. 64%, p < 0.02). The decrease of the infectious complications (4 vs. 12 cases, p < 0.005) could be possibly influenced by the increase of the nitrogen balance (12 ± 2 vs.
- 5 ± 1 g N, p < 0.05), as well as the levels of IgA (335.7 ± 31.44 vs. 357.81 ± 83.61 mg/dL, p = 0.029) and IgG between intervention and the control group [81].
S¸ahin et al. [88] reported a lessening in CRP in both groups, although the decrease was more pronounced in the study group (−38 vs. 18.6%, p = 0.00 and p = 0.01, respectively). However, these values were still higher than usual and a reason for this could be the presence of fewer leukocytes, and inflammation. In this trial, there was a reduction in LOS (14.2 ± 4.4 vs. 16.4 ± 3.9 days, p > 0.05) and complication rates (10 vs. 40%, p < 0.05) which may be due to an increase in CD4 and CD8 lymphocytes, although the latter failed to reach significance (p > 0.05). The increase in T-cell lymphocytes was also reported in other studies [80,83,87]. Nevertheless, in line with the latter, these measurements did not reach significance (p > 0.05).
Fuentes-Orozco et al. [93] reported a significant increase of nitrogen balance (p = 0.04), total lymphocytes count (p = 0.04), CD4 lymphocytes (p = 0.03), CD8 lymphocytes (p = 0.03), IgA (p = 0.01) and IL-10 (p = 0.02), an anti-inflammatory cytokine, were observed, along with a significant decrease of CRP (0.005) and IL-6 (p = 0.03). These data might explain the decrease in the incidence of infectious complications in the supplemented group (68.4%) when compared to the control group (31.6%, p = 0.03), as well as the mortality (9 vs. 22.7%), although the latter failed to reach significance (p = 0.20).
According to a review conducted by Novak et al. [54], the minimum concentration of glutamine to obtain positive clinical outcomes is 0.2 g/kg/day Gln, corresponding to 0.303 g/kg/day Ala-Gln dipeptide. Nevertheless, this amount could be raised to 20 g/day Gln according to Wischmeyer et al. [78], Heyland et al. [109] and García-De-Lorenzo [110]. Regarding Gln toxicity, Garlick [111] suggested that doses as high as 50–60 g/day Gln for several weeks were safe and showed no adverse effect. The use of a lower amount of glutamine could in part explain the lack of significance in the reduction of cytokines levels in some studies [69]. In the study by Engel et al. [63], dose requirements according to the previous studies [54,78,109,110] were met. In this case, a reason to explain these results may be the short time used for supplementation (3 days) rather than the dose used.
The form of supplementation used might also affect the outcome, even if the minimum amount of glutamine is given. This could be explained since glutamine in the form of
- L-Gln presents much lower stability than Ala-Gln dipeptide [78]. In addition, the effect of glutamine may also vary depending on whether it is given postoperatively and parenterally, and whether it is given preoperatively [95,104] and enterally [103], respectively. The observed beneficial effect on L/M ratio and TNFα levels was more pronounced when glutamine was administered by the enteral route. However, since the authors do not specify the amount of the administered glutamine, it is difficult to determine whether this observation is dose dependent or is due to the method of administration being the enteral route.
The shortest study [63] supplemented patients for just 3 days with 0.5 g/kg/day Gln in a double-blind RCT. In this trial, no significant differences were found in total lymphocytes count (p > 0.05), IL-6 levels (p < 0.05), LOS (2.6 ± 2.0 vs. 2.0 ± 1.7, p = 0.44), CRP (p = 0.72), IL-8 (p > 0.05) or TNFα (p > 0.05). This might suggest that a minimum length of 5 days is needed to achieve an effect in at least one of the parameters related to healing [69].
Besides, according to Morlion et al. [71], glutamine supplementation may also have a positive effect on the patient’s mood and general well-being. A reason for this could be the role of Gln as a neurotransmitter [112,113] as its depletion has been shown to cause mood disorders such as depression [113–115].
Therefore, it would be useful to measure this parameter in future studies to corroborate this effect.
5. Limitations of the Review
In the case of arginine, five studies were included in the meta-analysis. However, it was not possible to assess the effect of arginine supplementation on nitrogen balance and T-cell lymphocytes due to the lack of studies reporting these data. Besides, the studies had relatively small sample sizes. Therefore, more studies are required in this area of research. Besides, the high heterogeneity of the studies of both amino acids might have also affected the results of the meta-analysis.
6. Conclusions
This systematic review and meta-analysis have demonstrated that supplementation with either arginine and glutamine can positively influence wound healing or parameters related to healing including LOS and mortality. The effect of arginine supplementation was significant in relation to hydroxyproline content (p < 0.00001), while glutamine supplementation had significant effect on nitrogen balance (p < 0.0001), patient mortality (p = 0.0004), L/M ratio (p < 0.00001), LOS (p < 0.00001), CRP (p < 0.00001), IL-6 levels (p = 0.0001) and TNFα levels (p < 0.00001). However, the effect of glutamine supplementation on T-cell lymphocytes failed to reach significance (p = 0.07).
Author Contributions: Writing—original draft preparation, E.A.-L.; Writing—review and editing, N.Z.; O.O.; Supervision, N.Z.; Funding, M.J.S. and T.K. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding. Conflicts of Interest: The authors declare that there are no conflict of interest.
Figures
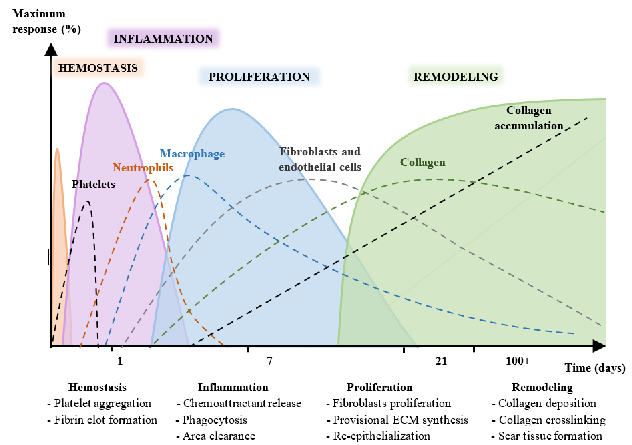
Figure 6
PRISMA flow diagram details the screening and selection process for studies on glutamine supplementation and wound healing. From the initial database search, studies were filtered through title screening, abstract review, and full-text assessment.
flowchart
Figure 7
Characteristics of included studies on arginine supplementation for wound healing are summarized in tabular form. The table captures study design, participant demographics, dosage protocols, and primary outcome measures across the selected trials.
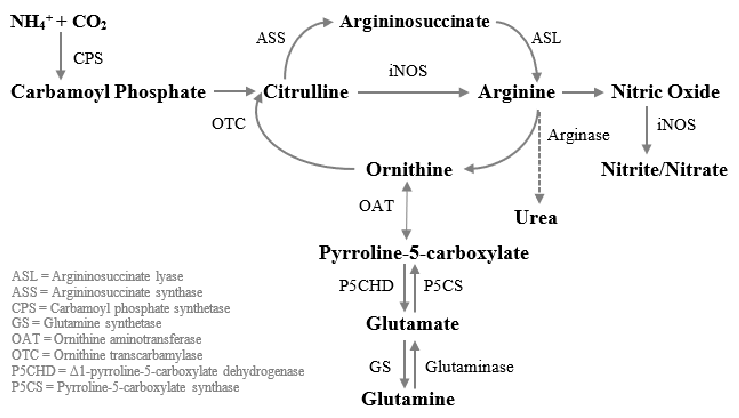
Figure 8
Glutamine-to-arginine metabolism in human macrophages is mapped, showing the conversion of carbamoyl phosphate and ornithine to citrulline via ornithine transcarbamylase, and subsequent transformation to argininosuccinate. This pathway is central to understanding how glutamine supports immune-mediated wound repair.
diagram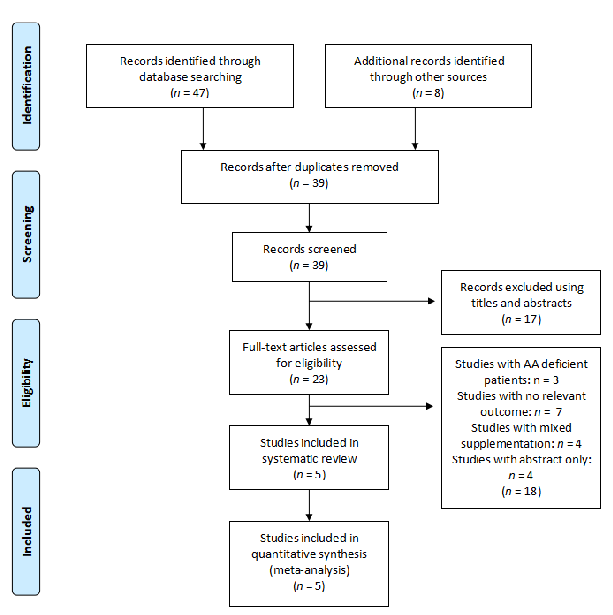
Figure 9
Study characteristics for the glutamine supplementation arm of the systematic review are presented. The compilation enables cross-study comparison of dosing regimens, wound types, and healing outcomes measured in human trials.
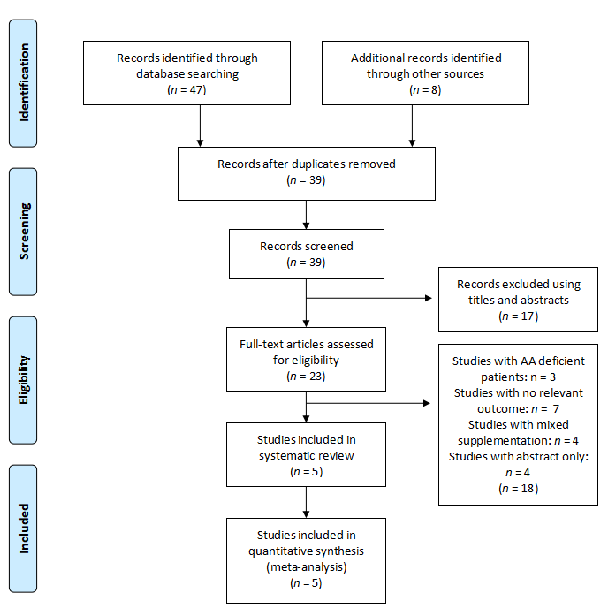
Figure 10
Risk of bias assessment for arginine studies is displayed using the Cochrane tool framework. Each domain of potential bias is evaluated across the included trials to gauge the overall quality of evidence supporting arginine supplementation for wound healing.
chart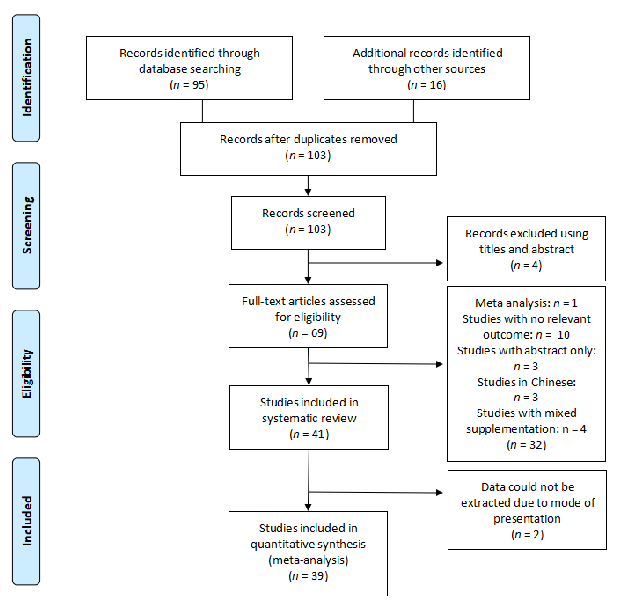
Figure 11
Risk of bias assessment for glutamine studies complements the arginine evaluation. The systematic assessment of selection, performance, detection, attrition, and reporting bias helps contextualize the strength of conclusions about glutamine and wound repair.
chart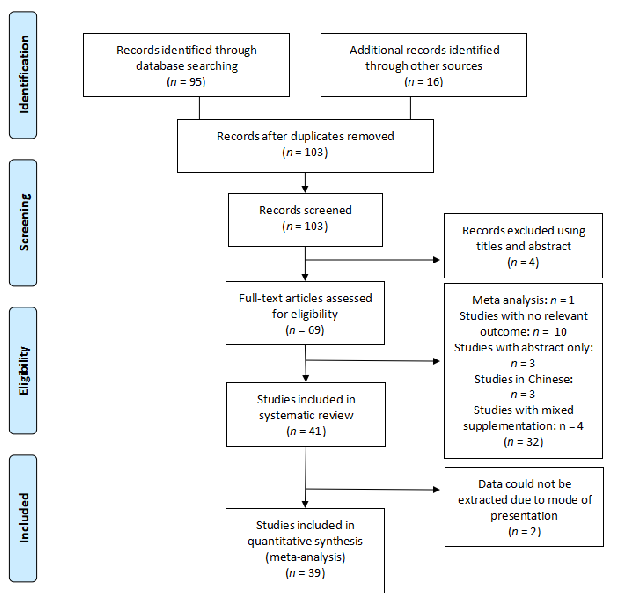
Figure 12
The PRISMA flow diagram for the arginine search strategy traces article identification through database queries, duplicate removal, and eligibility screening. Five human studies on arginine supplementation and wound healing met the final inclusion criteria.
flowchart
Figure 13
Risk of bias is summarized for each included arginine study using green (low risk), yellow (unclear), and red (high risk) indicators. The visual matrix helps readers quickly assess methodological quality across randomization, blinding, and outcome reporting domains.
chart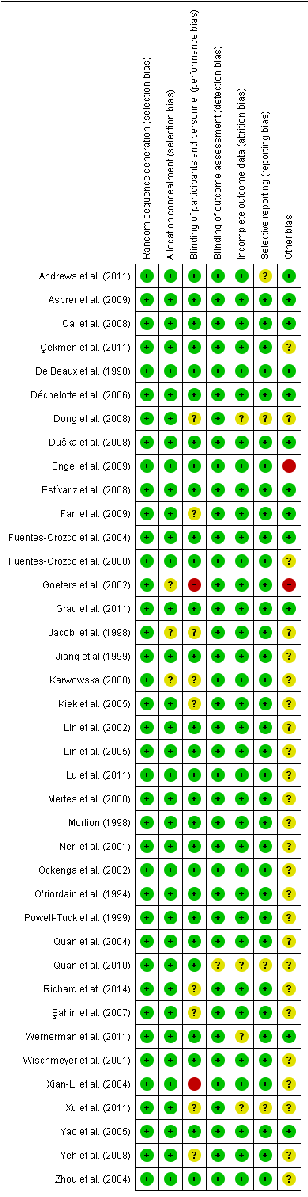
Figure 14
Risk of bias for the included glutamine studies follows the same Cochrane framework, with symbols indicating low, unclear, and high risk across multiple domains. The assessment covers 39 human studies evaluating glutamine supplementation effects on wound healing.
chart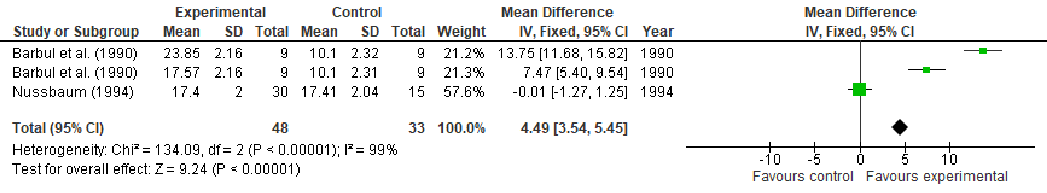
Figure 15
A fixed-effects meta-analysis forest plot displays the effect of arginine supplementation on hydroxyproline content, a biomarker of collagen synthesis in wound tissue. Individual study effect sizes and confidence intervals are pooled to estimate the overall treatment effect.
forest_plot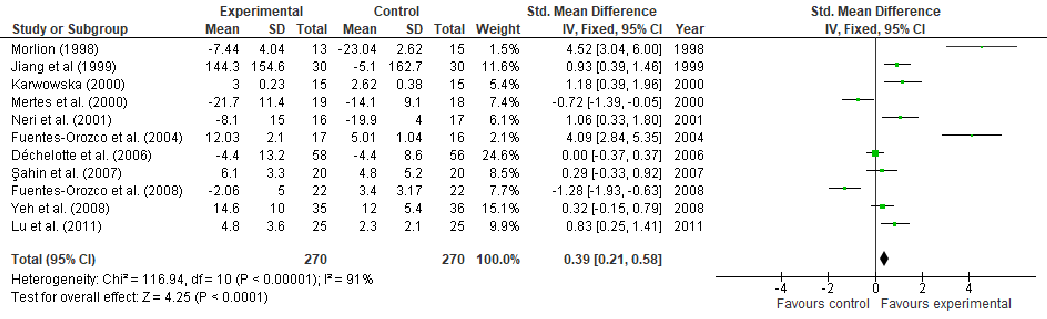
Figure 16
Nitrogen balance outcomes from glutamine supplementation studies are combined in a fixed-effects forest plot. Positive nitrogen balance indicates enhanced protein retention, which is associated with improved wound healing capacity in stressed or surgical patients.
forest_plot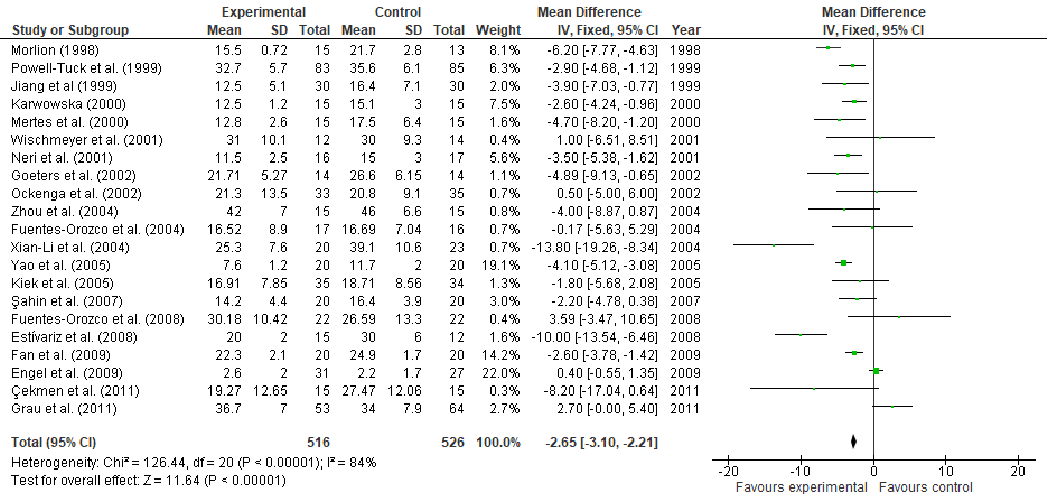
Figure 17
Length of hospital stay is analyzed in a fixed-effects meta-analysis forest plot for glutamine supplementation trials. The pooled estimate indicates whether glutamine may reduce recovery time in patients with wounds requiring hospitalization.
forest_plot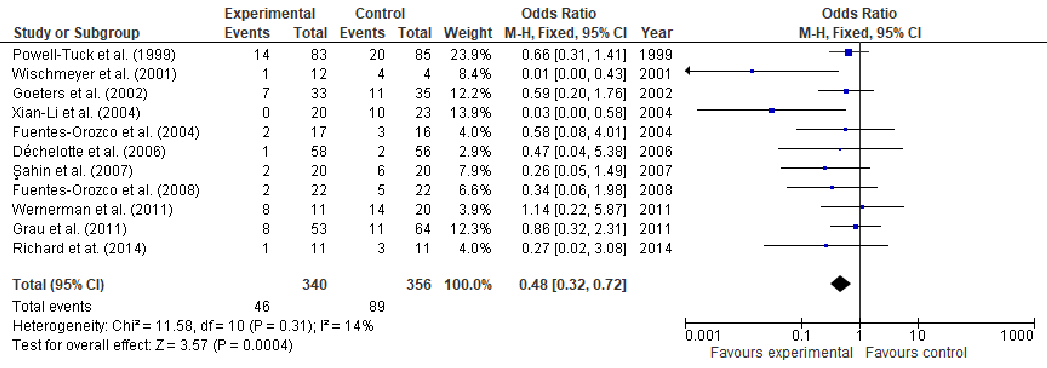
Figure 18
Additional meta-analysis results for glutamine supplementation and wound healing outcomes are presented. The analysis extends the evidence base by examining secondary endpoints across the included clinical trials.
forest_plot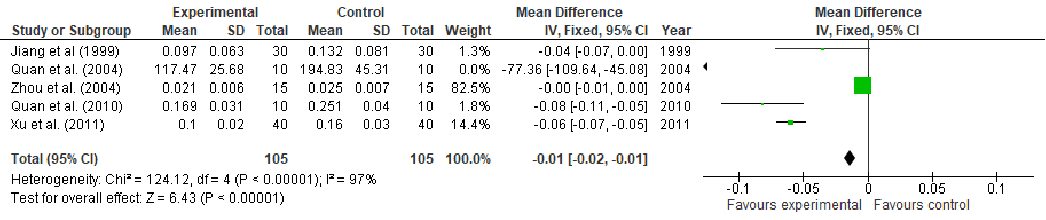
Figure 19
Supplementary forest plot data from the glutamine meta-analysis displays effect estimates for an additional wound healing outcome. Confidence intervals for individual studies and the pooled effect are shown.
forest_plot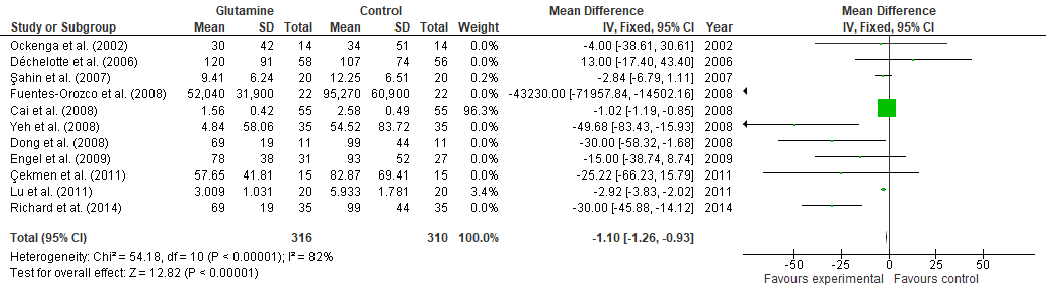
Figure 20
C-reactive protein levels following glutamine supplementation are pooled in a fixed-effects forest plot. Lower CRP values suggest reduced systemic inflammation, which may contribute to improved wound healing outcomes.
forest_plot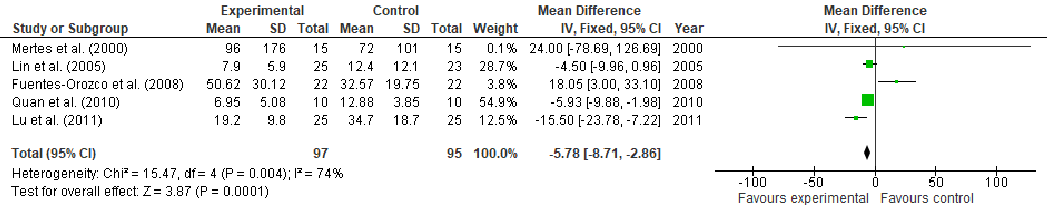
Figure 21
An additional inflammatory or immune marker from the glutamine meta-analysis is displayed in forest plot format. The pooled analysis evaluates whether glutamine supplementation modulates immune parameters relevant to wound repair.
forest_plot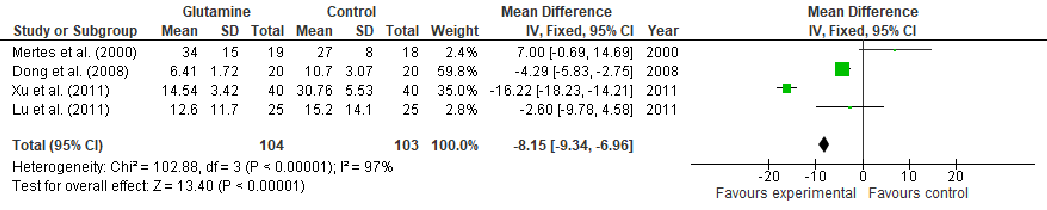
Figure 22
Further meta-analysis results present pooled effect sizes for wound healing parameters in glutamine-supplemented patients. The forest plot format allows visual comparison of effect magnitude and consistency across trials.
forest_plot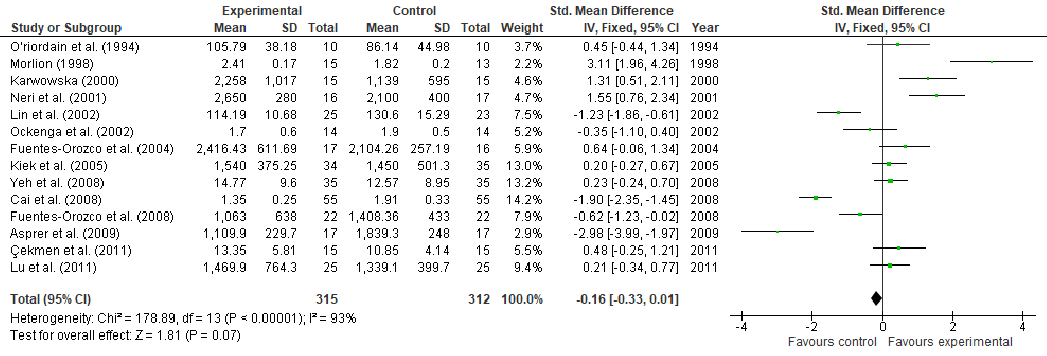
Figure 23
T-cell lymphocyte counts are analyzed in a fixed-effects forest plot across glutamine supplementation trials. Enhanced T-cell proliferation is associated with improved immune-mediated wound repair, and the pooled estimate evaluates glutamine's immunomodulatory potential.
forest_plotUsed In Evidence Reviews
Similar Papers
Critical reviews in immunology · 2012
M1 and M2 Macrophages: Oracles of Health and Disease.
Wound repair and regeneration : official publication of the Wound Healing Society [and] the European Tissue Repair Society · 2003
Arginine physiology and its implication for wound healing.
International journal of molecular sciences · 2019
Nutrition and Wound Healing: An Overview Focusing on the Beneficial Effects of Curcumin.
Nutrition in clinical practice : official publication of the American Society for Parenteral and Enteral Nutrition · 2005
Arginine supplementation and wound healing.
Current opinion in clinical nutrition and metabolic care · 2008
Assessment and nutritional aspects of wound healing.
ACS nano · 2023