Early intervention with Kan Jang® to treat upper-respiratory tract infections: A randomized, quadruple-blind study.
Study Design
- Study Type
- Other
- Sample Size
- 179
- Population
- None
- Intervention
- Early intervention with Kan Jang® to treat upper-respiratory tract infections: A randomized, quadruple-blind study. 60 mg
- Comparator
- placebo
- Primary Outcome
- None
- Effect Direction
- Positive
- Risk of Bias
- Unclear
Abstract
BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, called Kan Jang®, are traditionally used for relieving symptoms of upper-respiratory tract infections (URTIs). This study aimed to assess the efficacy of early intervention with Kan Jang® on the relief and duration of inflammatory symptoms during the acute phase of the disease. EXPERIMENTAL PROCEDURE: A total of 179 patients with URTI symptoms received six Kan Jang® (daily dose of andrographolides: 60 mg) or placebo capsules a day for five consecutive days in this randomized, quadruple-blinded, placebo-controlled, two-parallel-group phase II study. The primary efficacy outcomes were the decrease in the acute-phase duration and the mean URTI symptoms score (sore throat, runny nose, nasal congestion, hoarseness, cough, headache, and fatigue). RESULTS: Early intervention with Kan Jang® significantly increased the recovery rate and reduced the number of sick leave days by >21% (0.64/day) relative to that observed in the placebo group (2.38 vs. 3.02 days, p = 0.0053). Kan Jang® significantly alleviated all URTI symptoms starting from the second day of treatment. A superior anti-inflammatory effect of Kan Jang® to that of placebo was also observed on the white blood cell count (p = 0.007) and erythrocyte sedimentation rate (p = 0.0258). Treatment with Kan Jang® was tolerated well. CONCLUSION: This study demonstrates that early intervention with Kan Jang® capsules reduces the recovery duration of patients by 21% and significantly relieves the severity of typical URTI symptoms.
TL;DR
It is demonstrated that early intervention with Kan Jang® capsules reduces the recovery duration of patients by 21% and significantly relieves the severity of typical URTI symptoms.
Full Text
Name of the medicinal product
KanJang Andrographis® with fixed combination of Andrographis paniculata herb extract and Eleutherococcus senticosus root extract.
Pharmaceutical form
Capsules
Indication for use
Herbal Medicinal Product for the “reduction in the severity and duration of symptoms of uncomplicated respiratory tract infections (common cold)”.
Dosage The recommended daily dose is 2 capsules 3 times per day for adults.
Quantitative Composition
Substance Quantity per one capsule, mg
Active ingredients Herba Andrographidis standardised extract containing 66-110 mg of native dry extract ( DER 4,5-8,0 :1, extraction agent - ethanol* 70 %, V/V) corresponding to 10 mg of the sum of andrographolisdes (andrographolide and deoxydidehydro-andrographolide) and 45-67% of maltodextrin**(adjuvant for adjustment)
250
Radix Eleutherococci root extract, containing 11.4 mg of native dry extract (DER 17-30:1, extraction agent – ethanol 70%, V/V) , corresponding to 194-342 mg of herbal substance (Radix Eleutheorcocci, standardized for the content of eleutherosides B and E (0.4 mg/capsule) and 50% of maltodextrine** (adjuvant for adjustment )
22,8
Inactive ingredients Microcrystalline cellulose Magnesium stearate Vegetable capsule
70 7.2 75
Total weight 425 ± 21
Product: Kan Jang Andrographis Product category: Herbal medicinal product Dosage form: capsules
Daily dose: 6 capsules size 1 Batch number: 50145 Batch size: 5000 capsules
Composition
Product: Placebo Product category: Herbal medicinal product Dosage form: capsules Daily dose: 6 capsules size 1 Batch number: 50137-1 Batch size: 4000 capsules
Composition
Figures
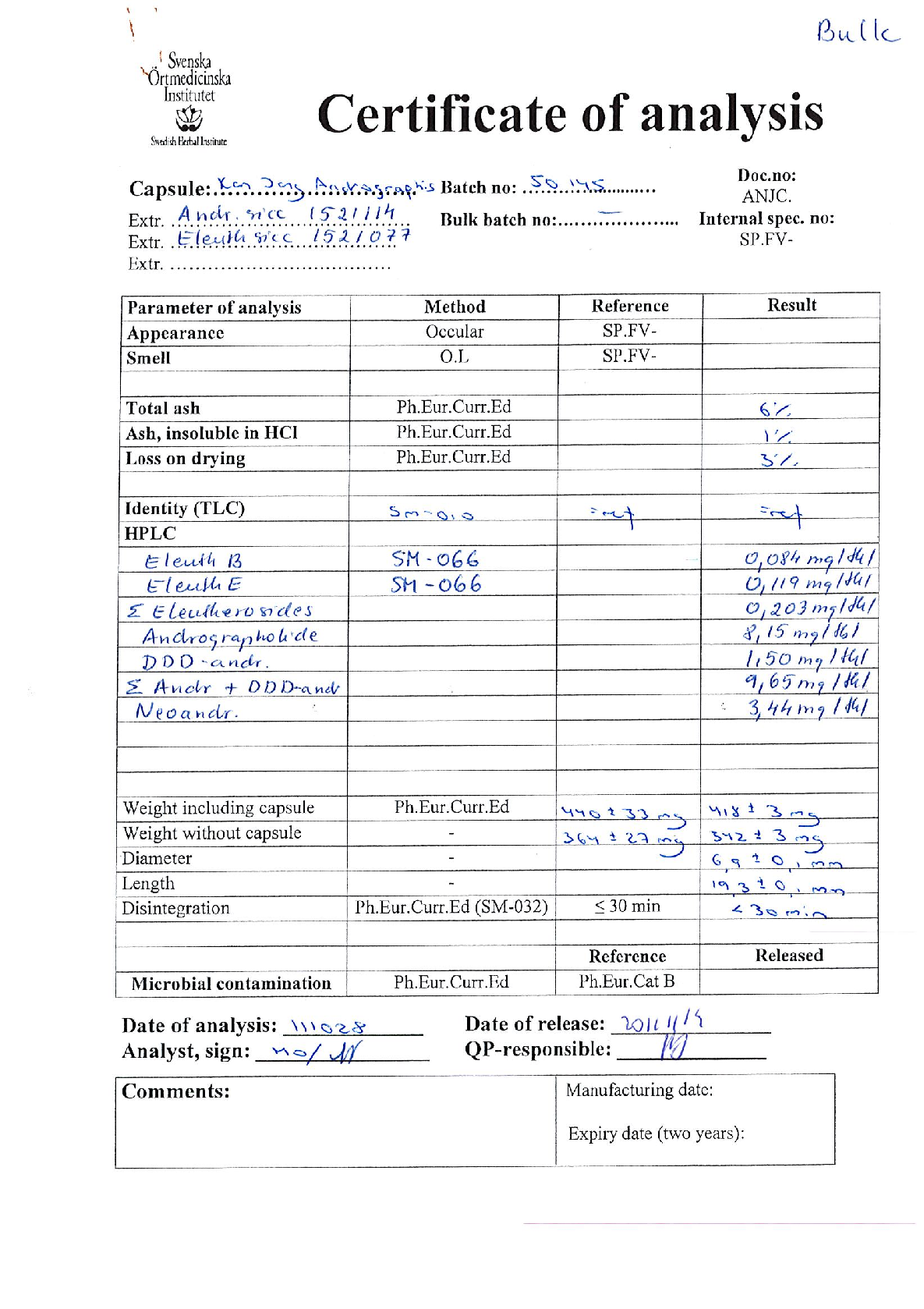
Figure 1
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 2
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart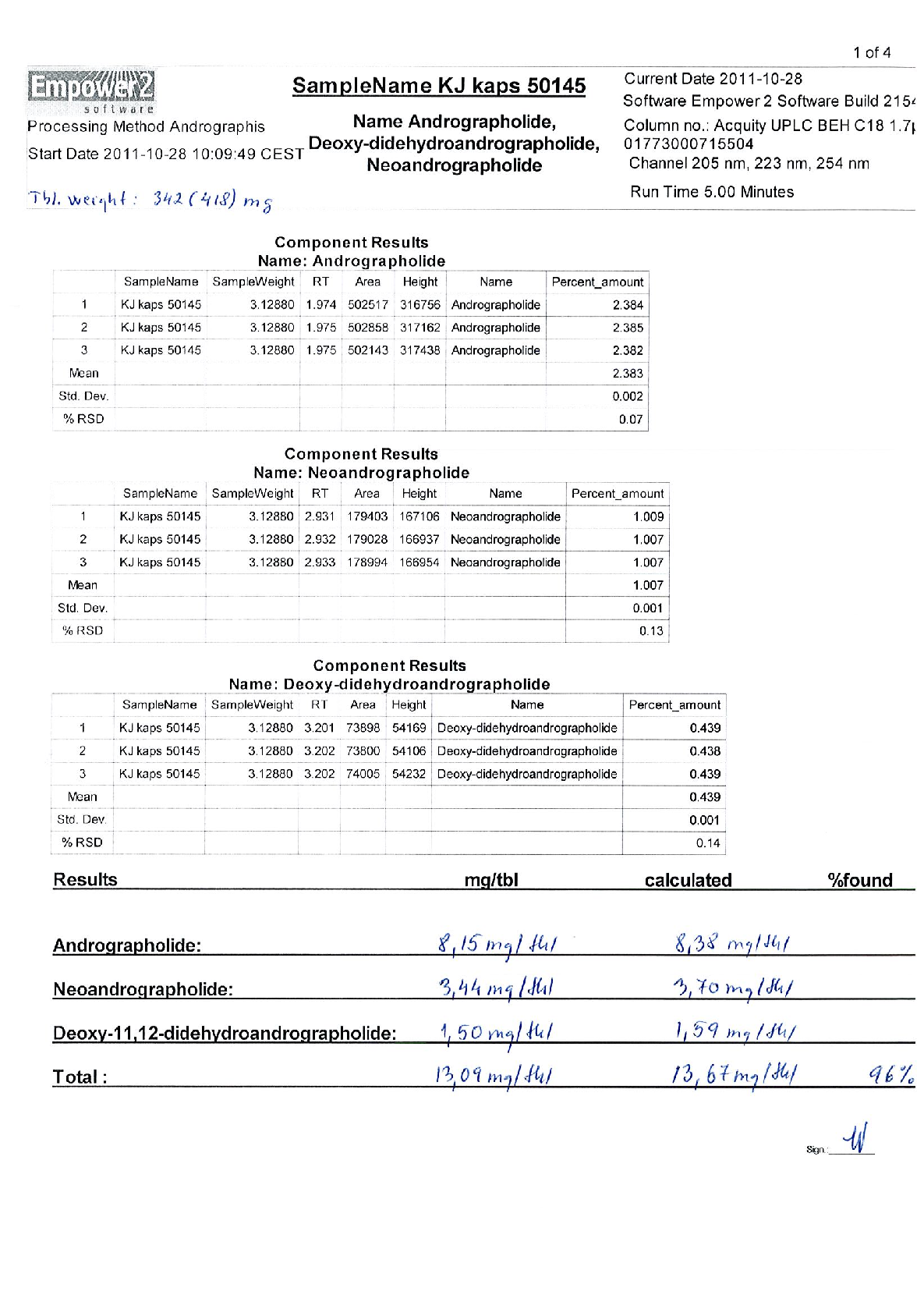
Figure 3
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart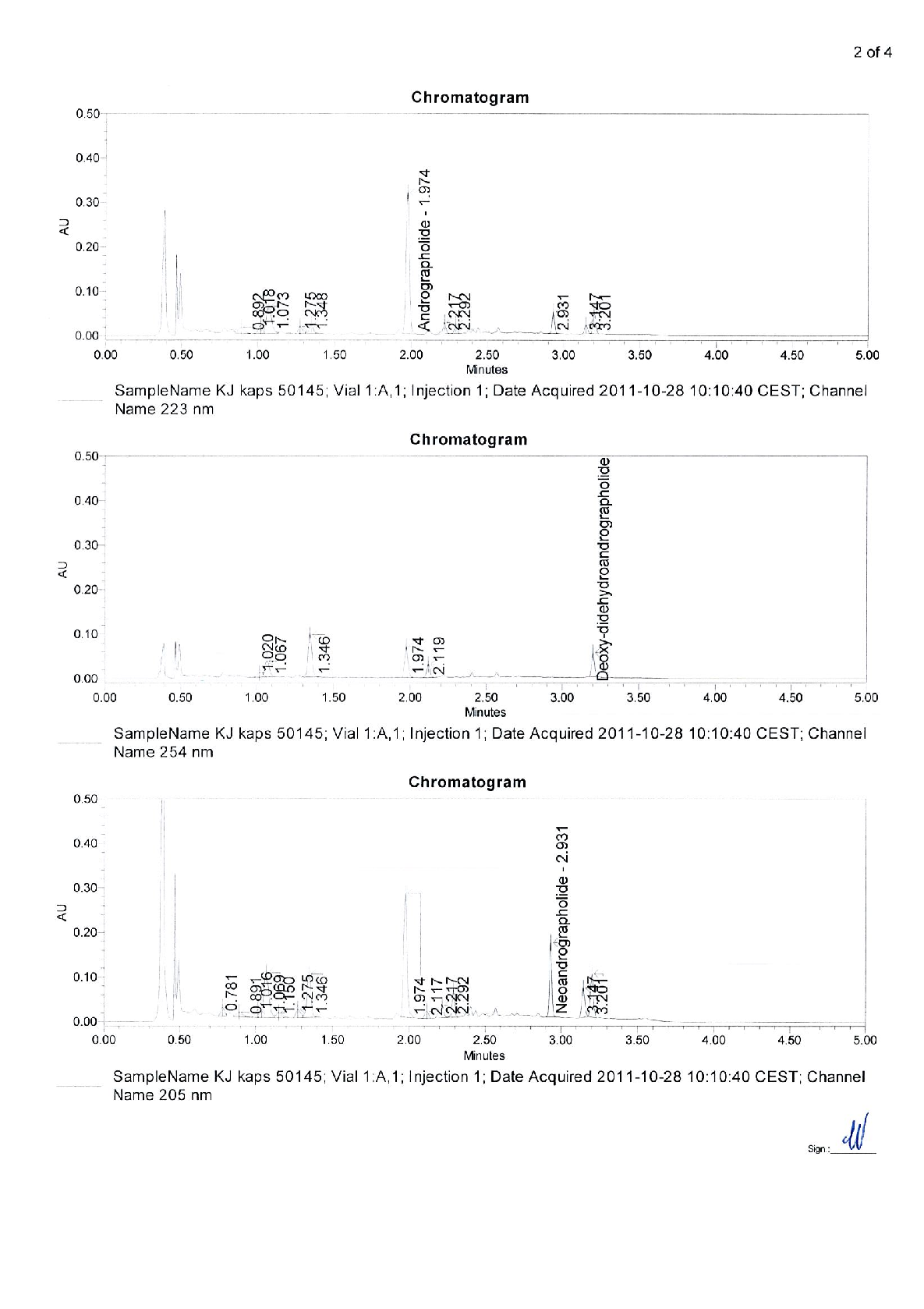
Figure 4
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart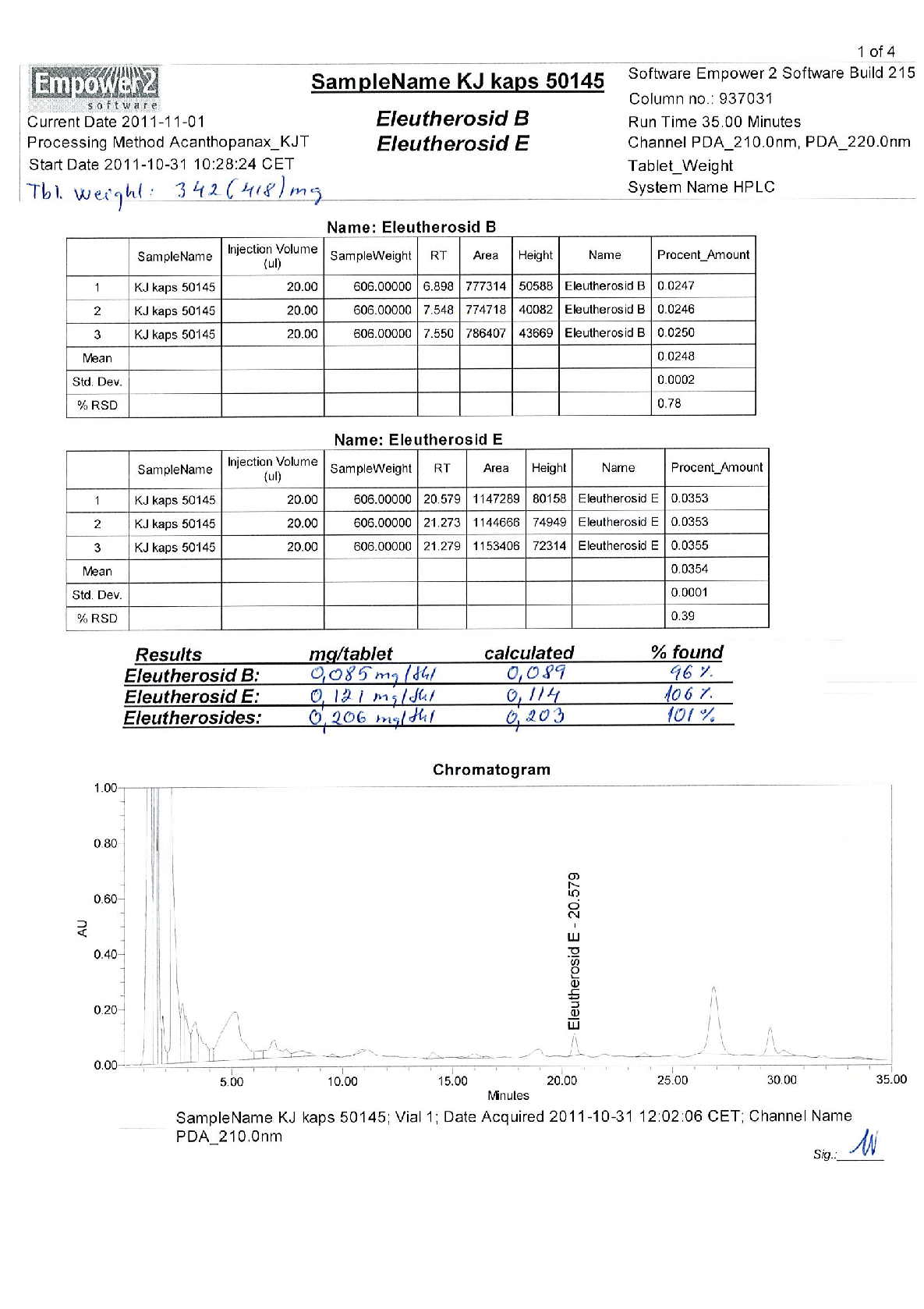
Figure 5
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart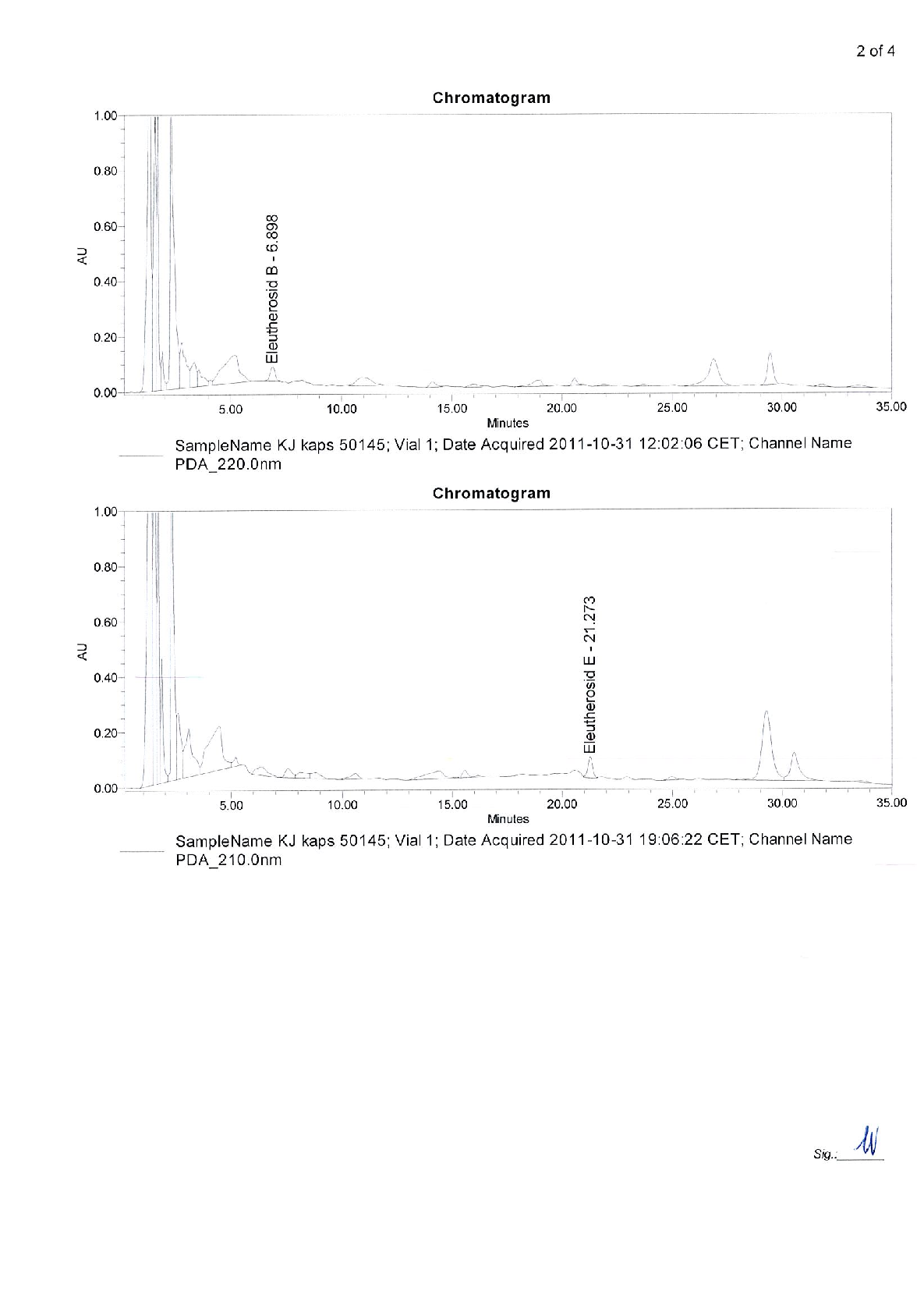
Figure 6
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart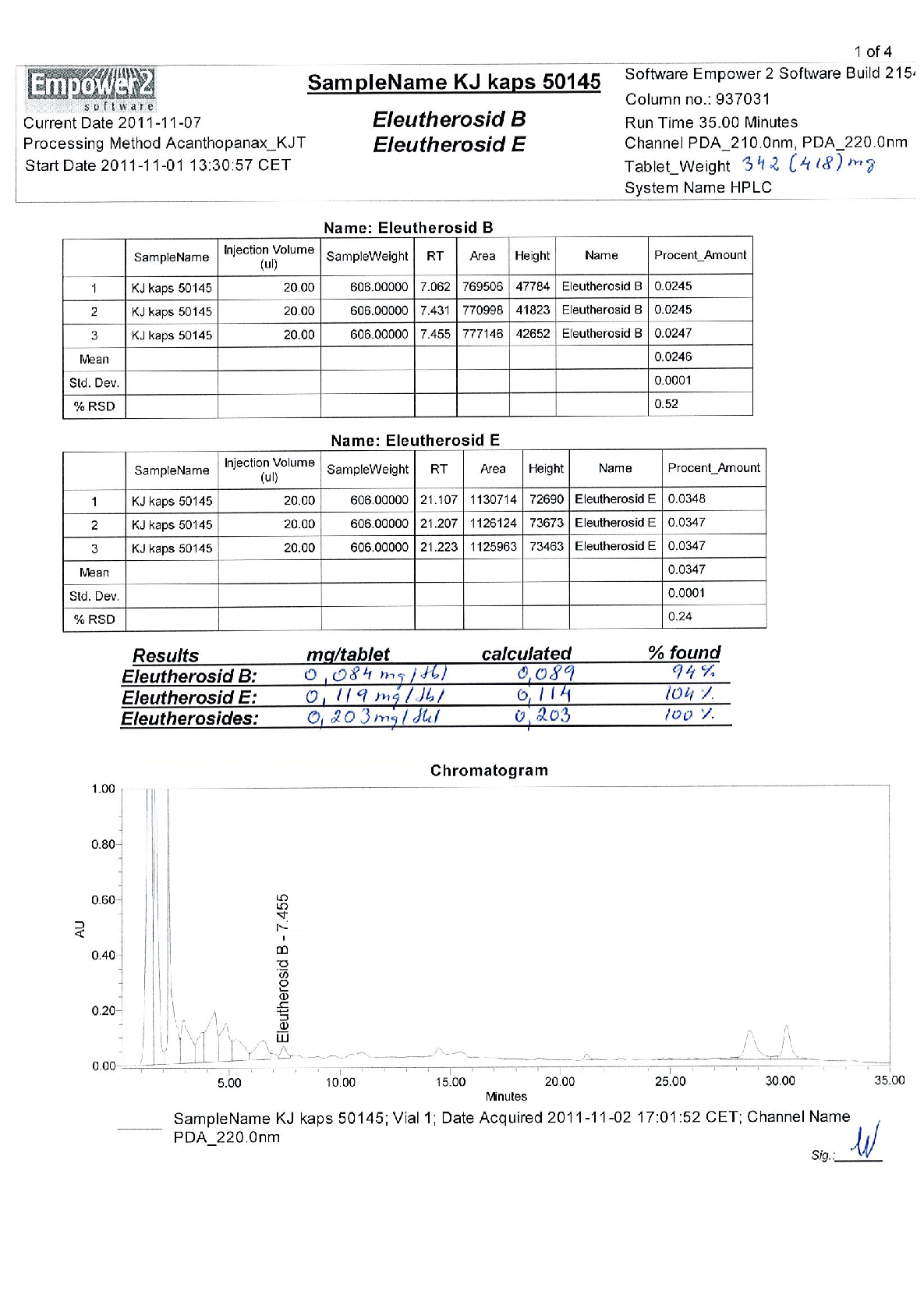
Figure 7
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart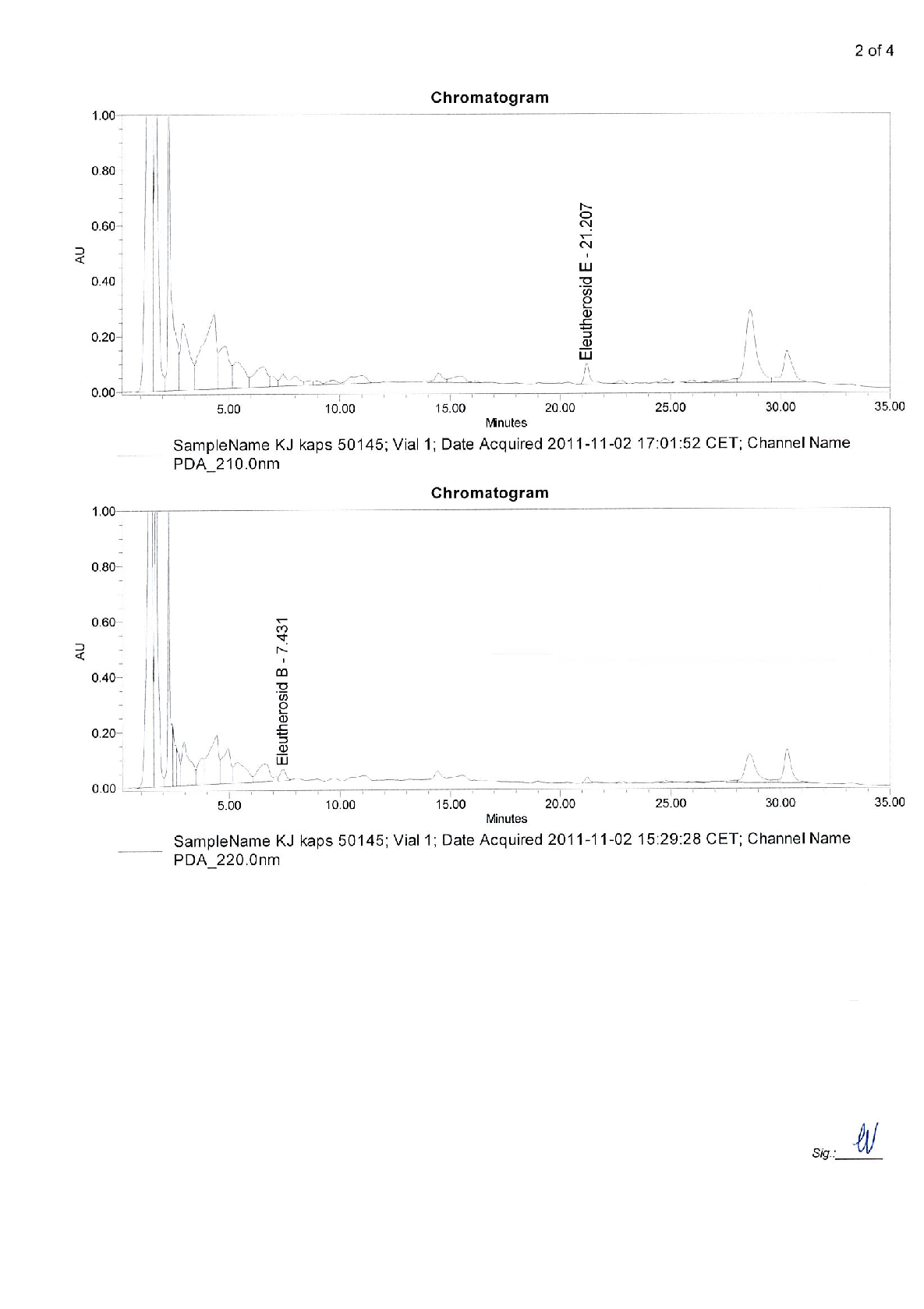
Figure 8
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart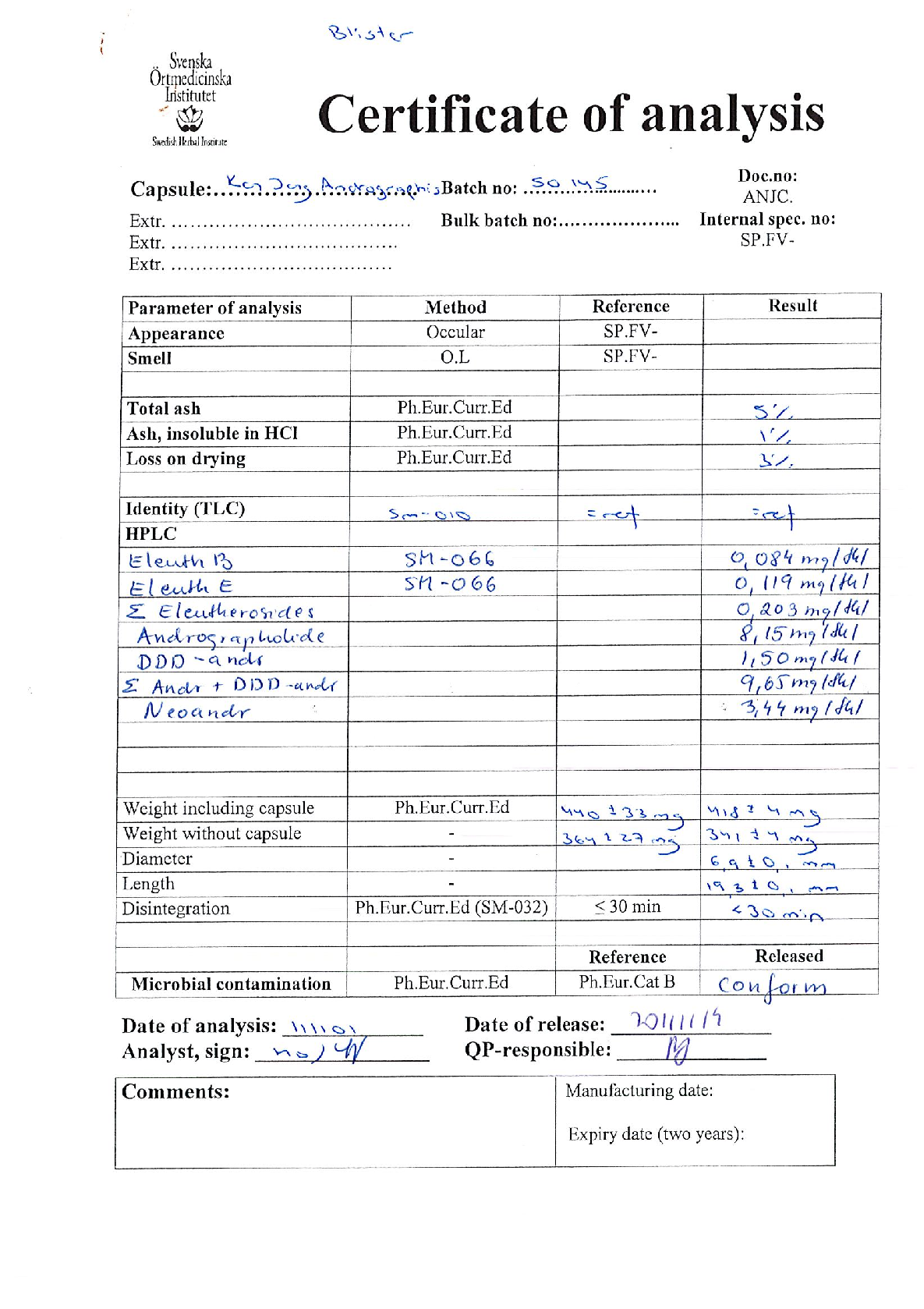
Figure 9
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart
Figure 10
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 11
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 12
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart
Figure 13
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 14
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart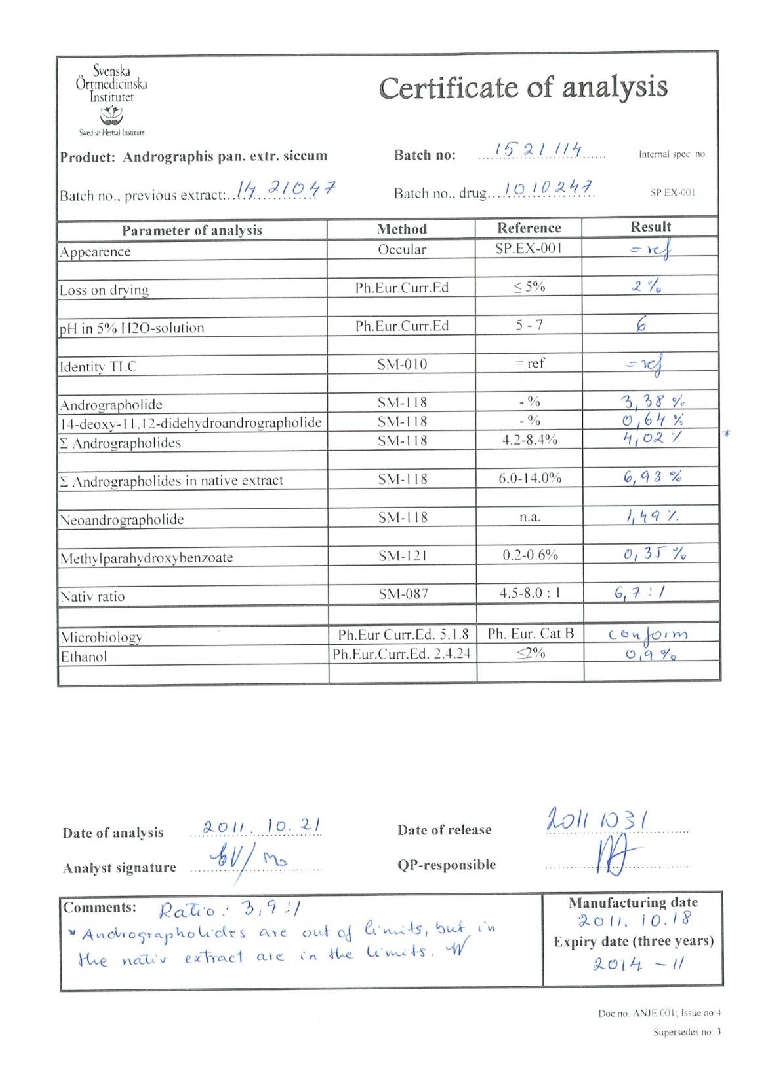
Figure 15
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart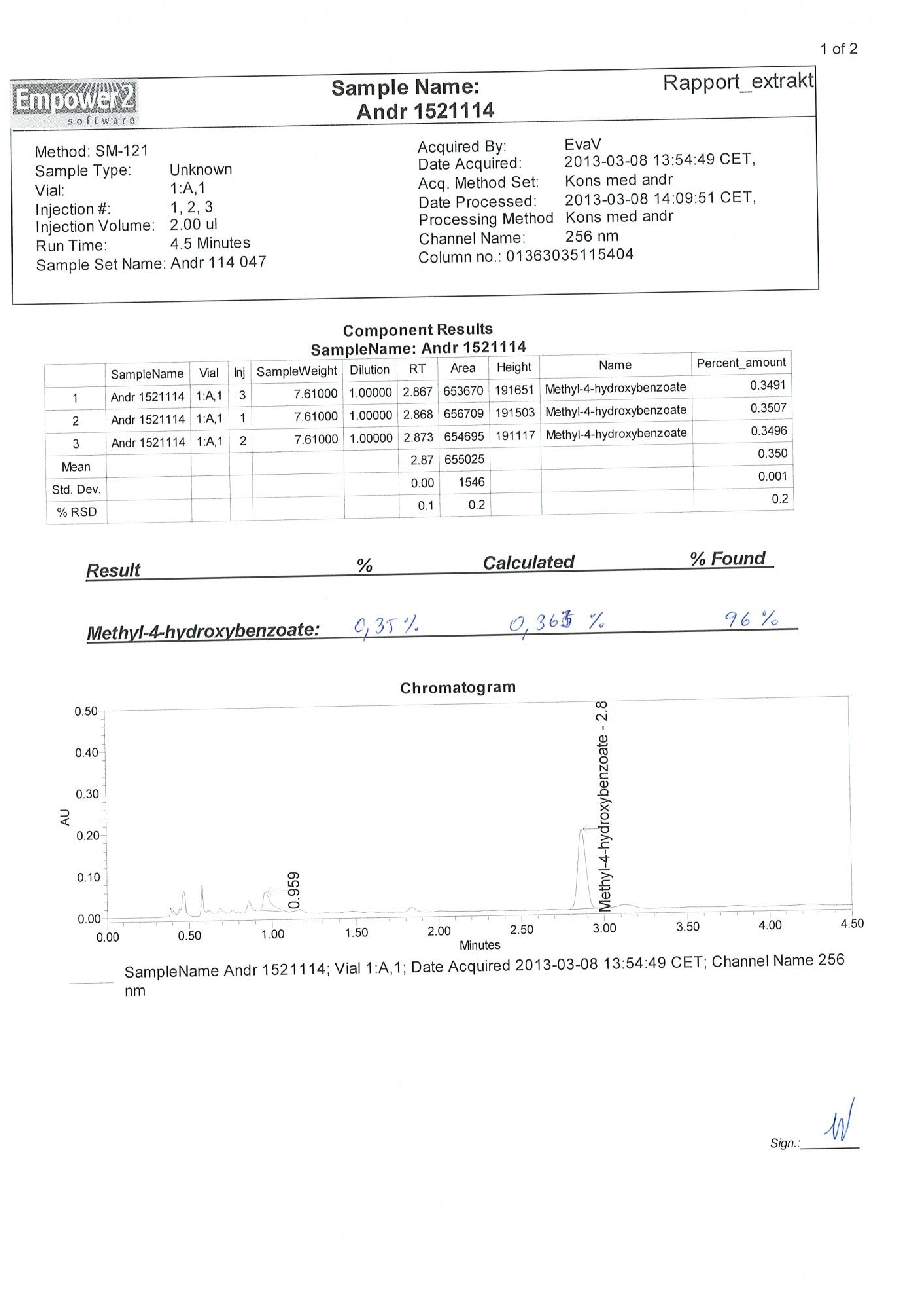
Figure 16
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 17
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart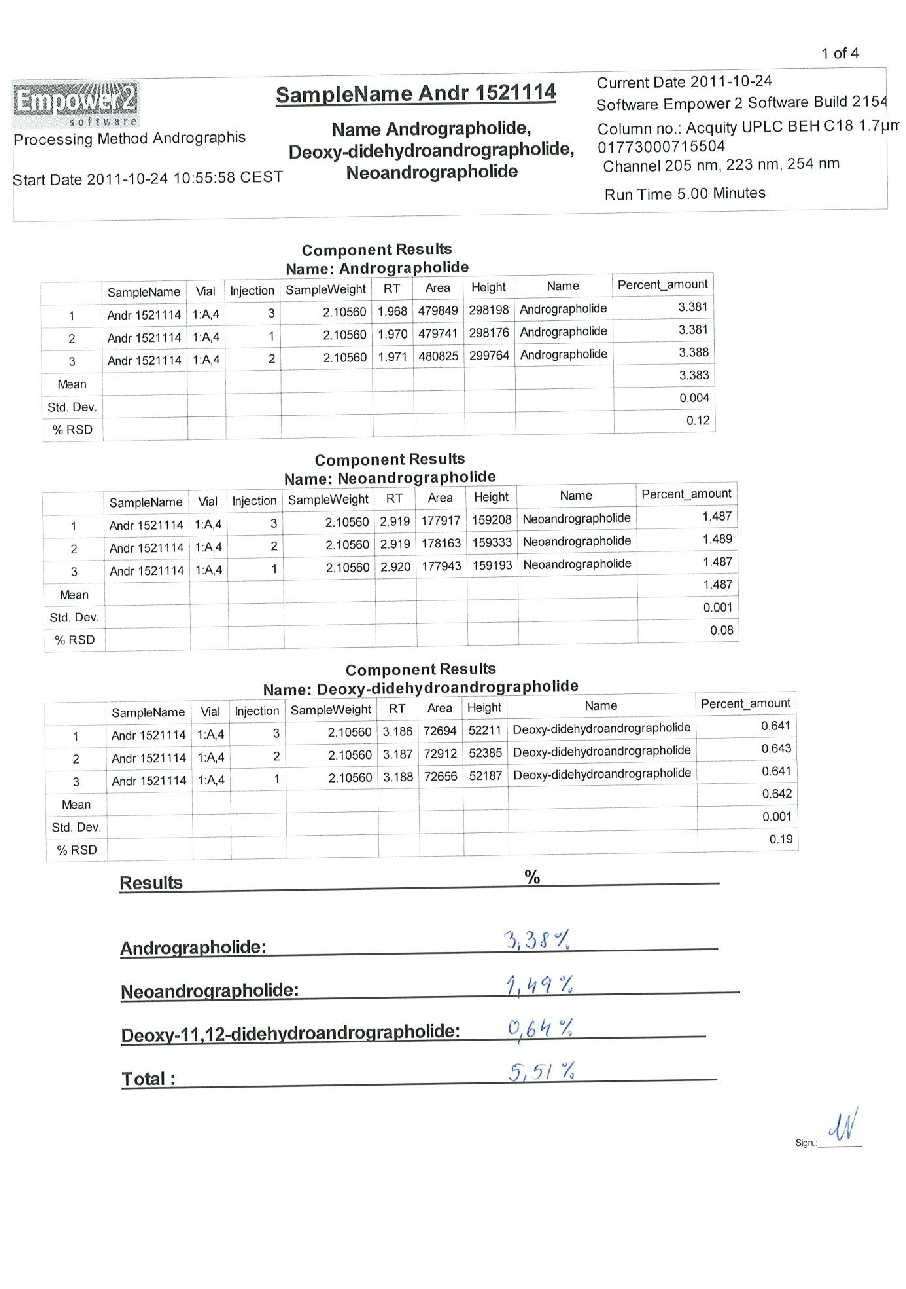
Figure 18
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart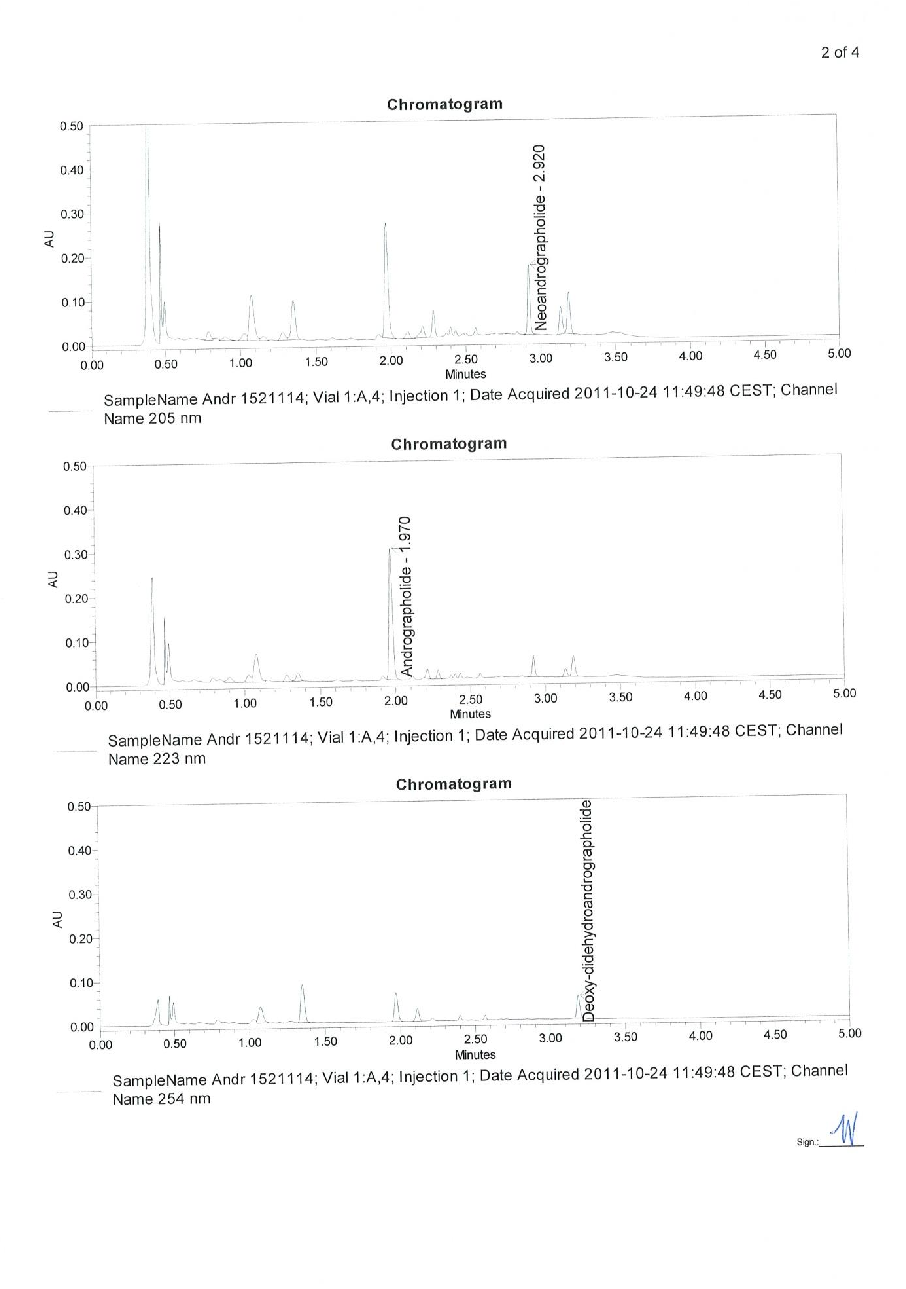
Figure 19
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart
Figure 20
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 21
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 22
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart
Figure 23
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 24
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart
Figure 25
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 26
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart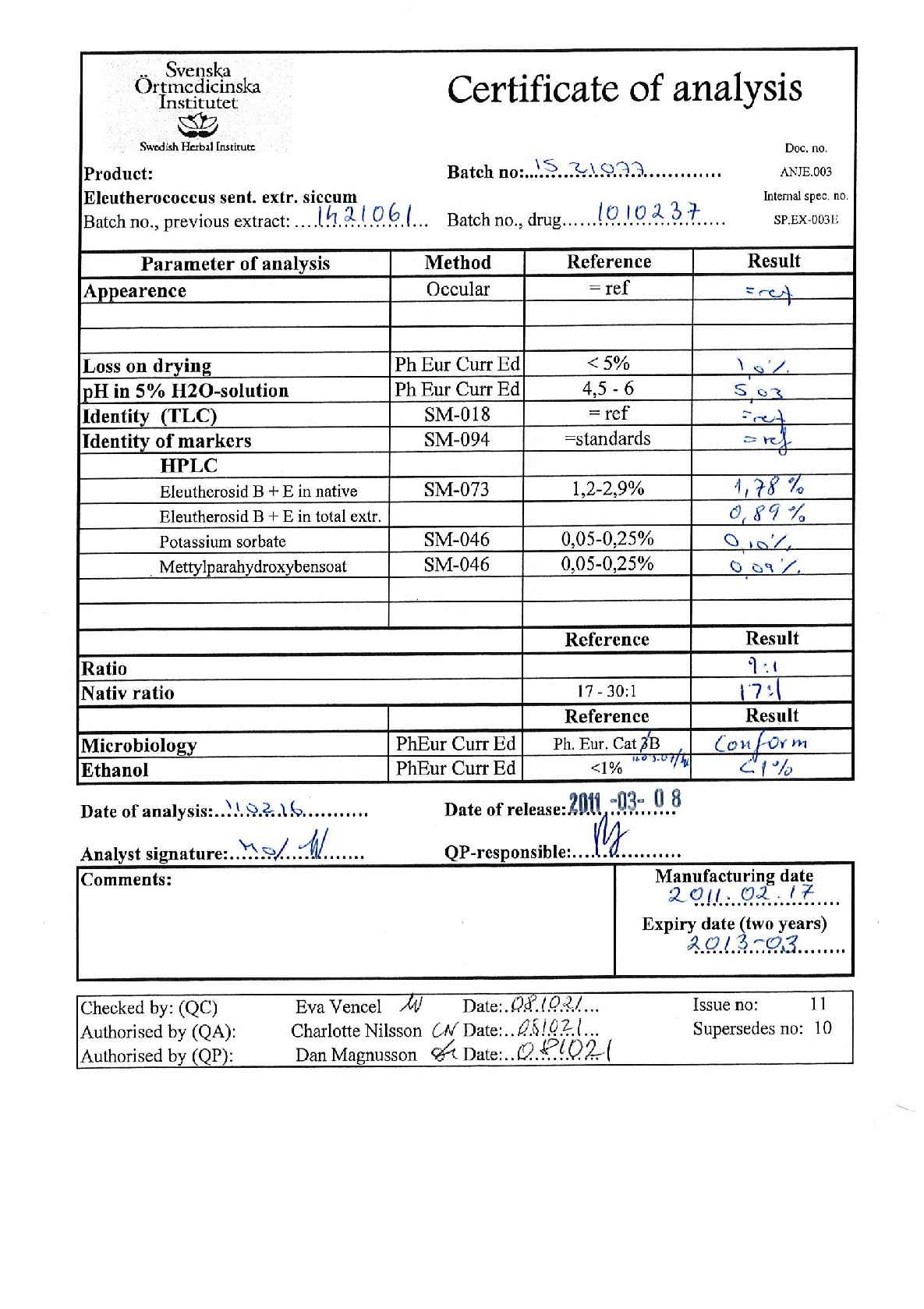
Figure 27
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart
Figure 28
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart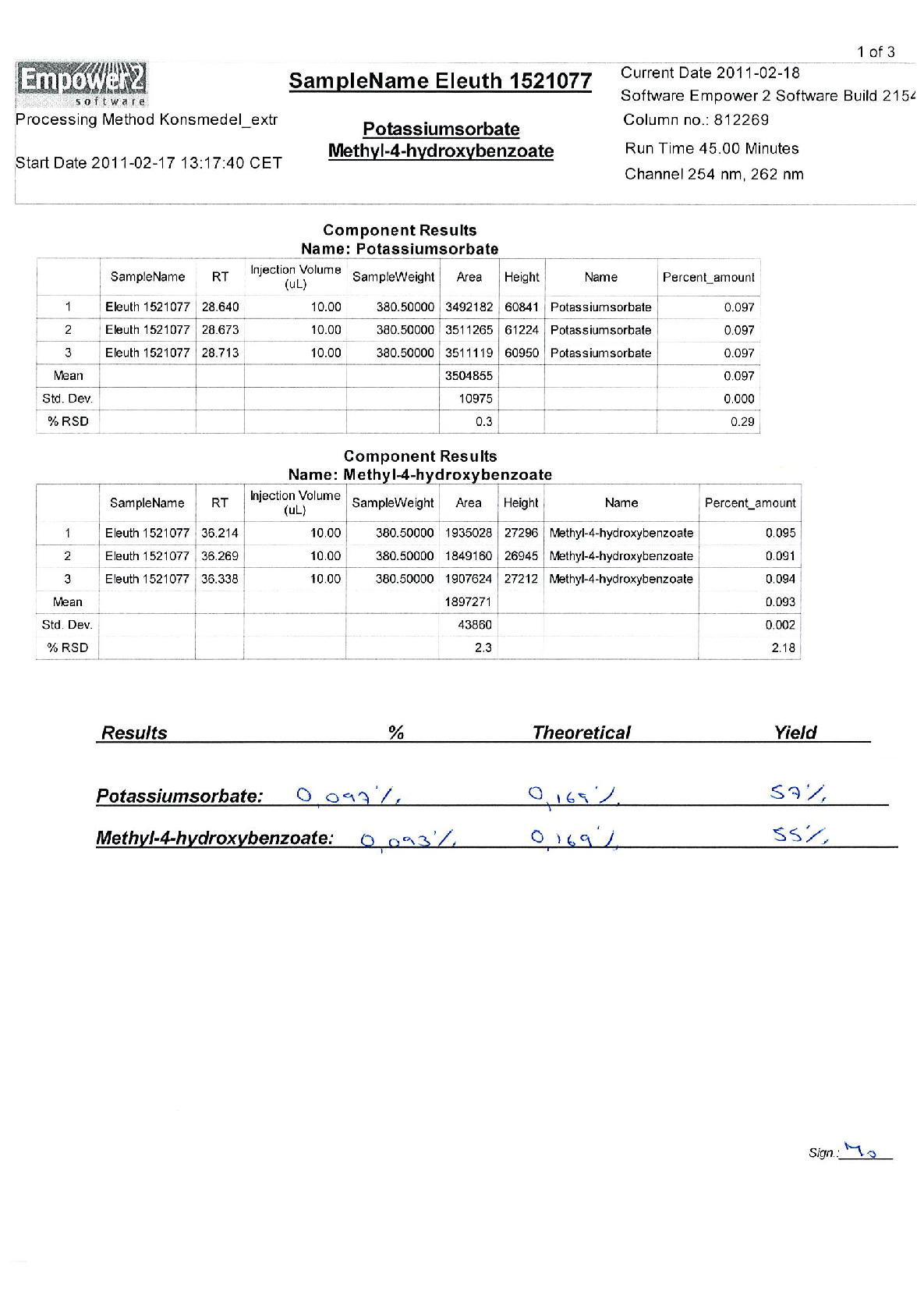
Figure 29
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart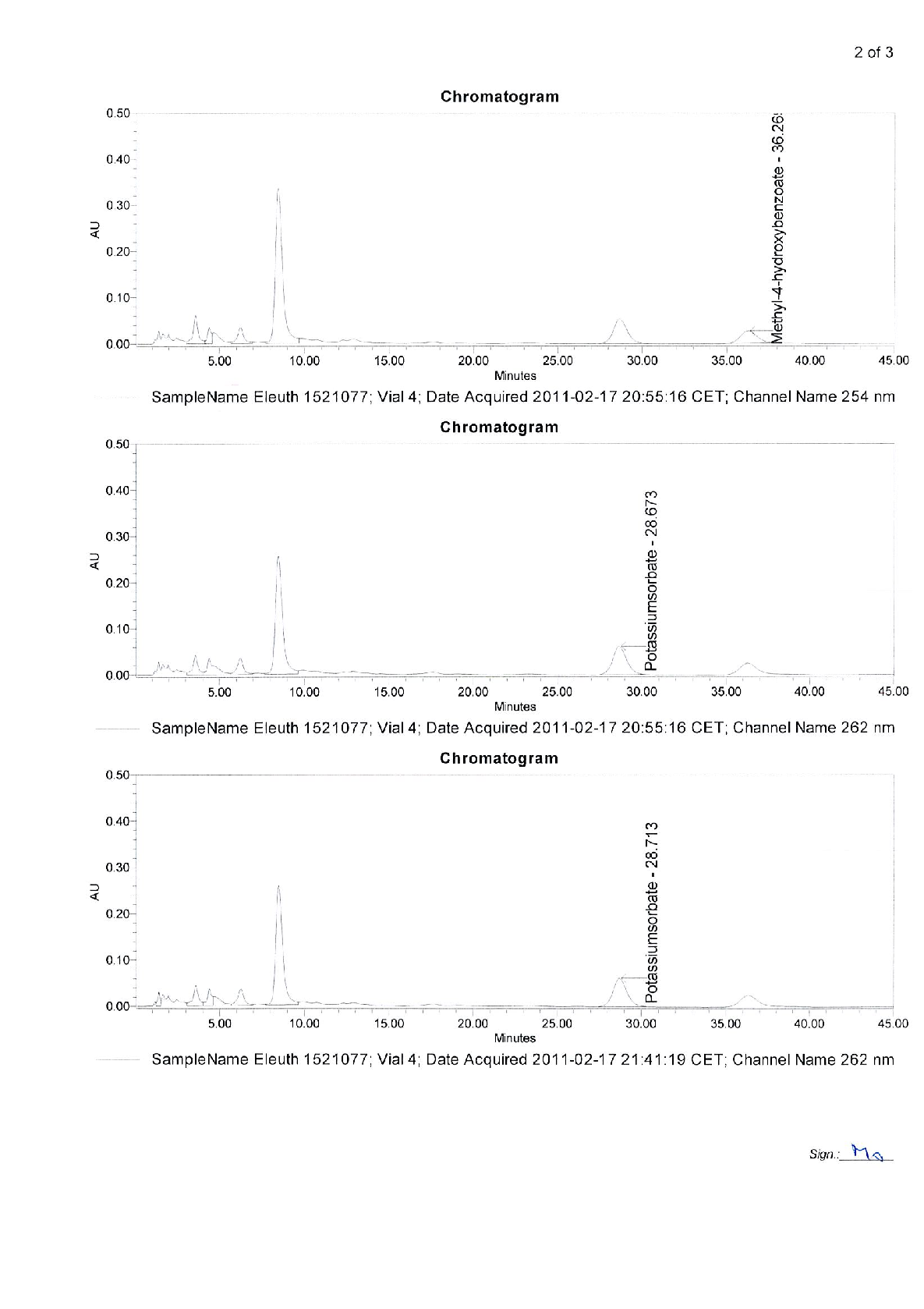
Figure 30
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart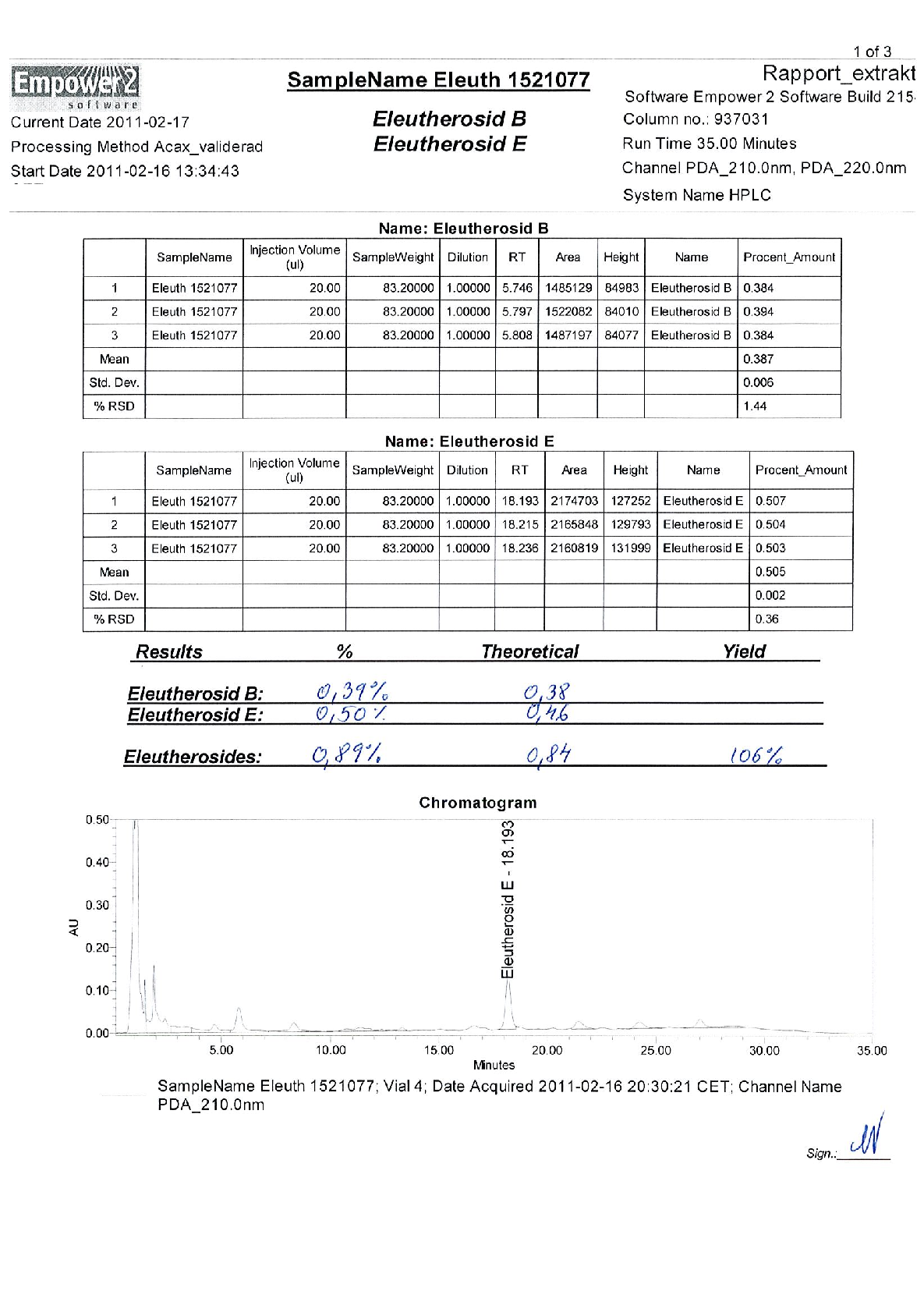
Figure 31
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart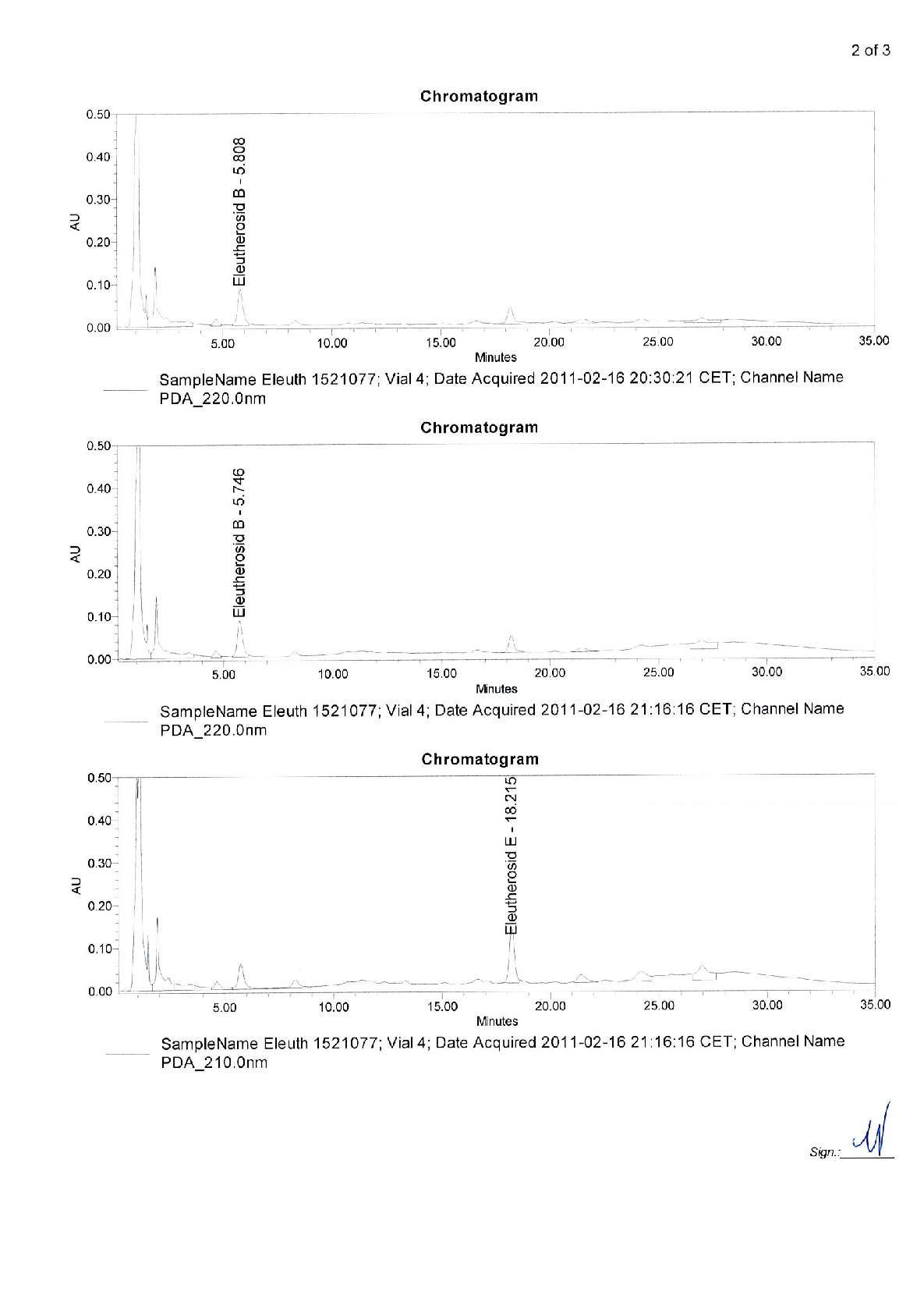
Figure 32
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart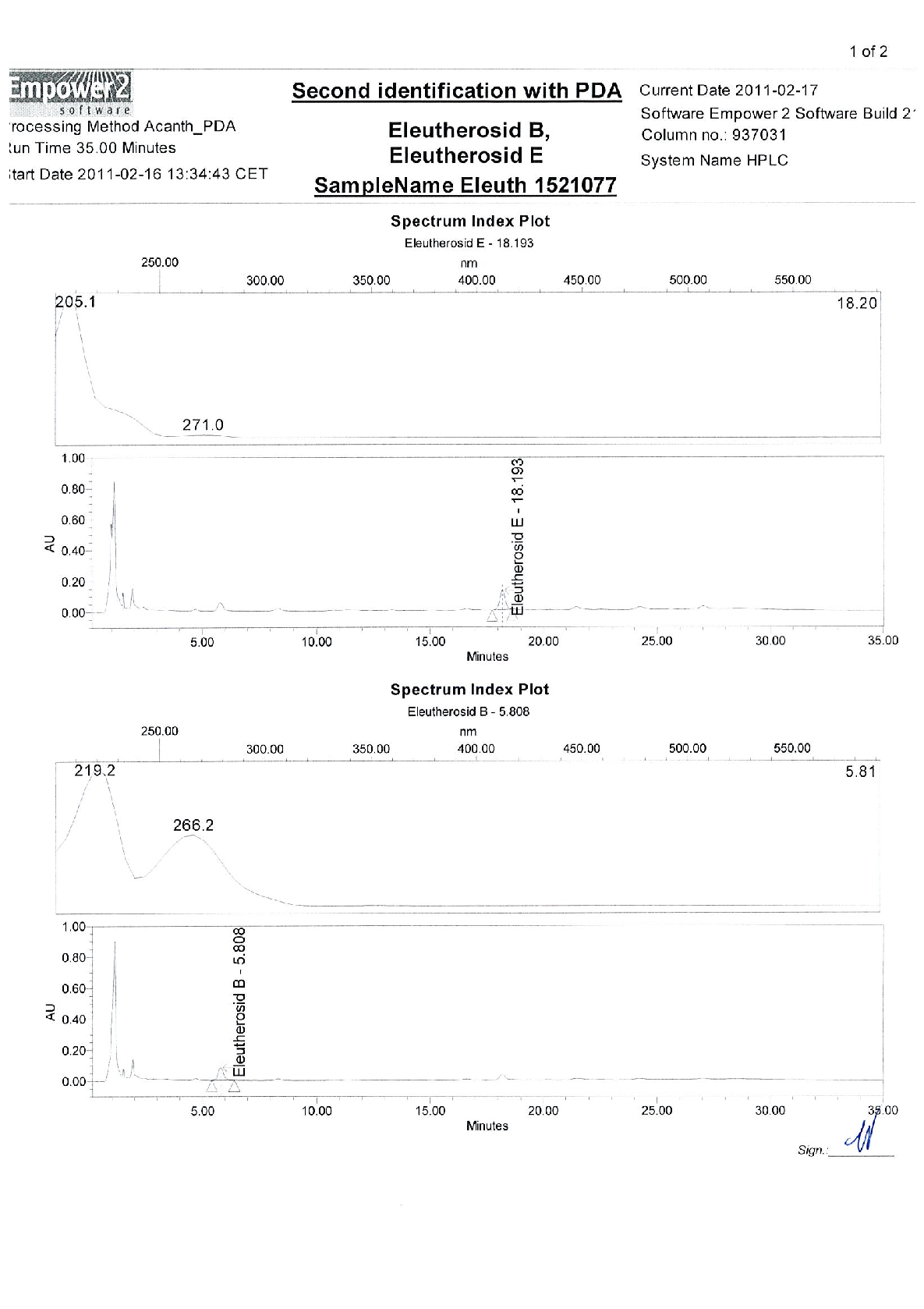
Figure 33
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart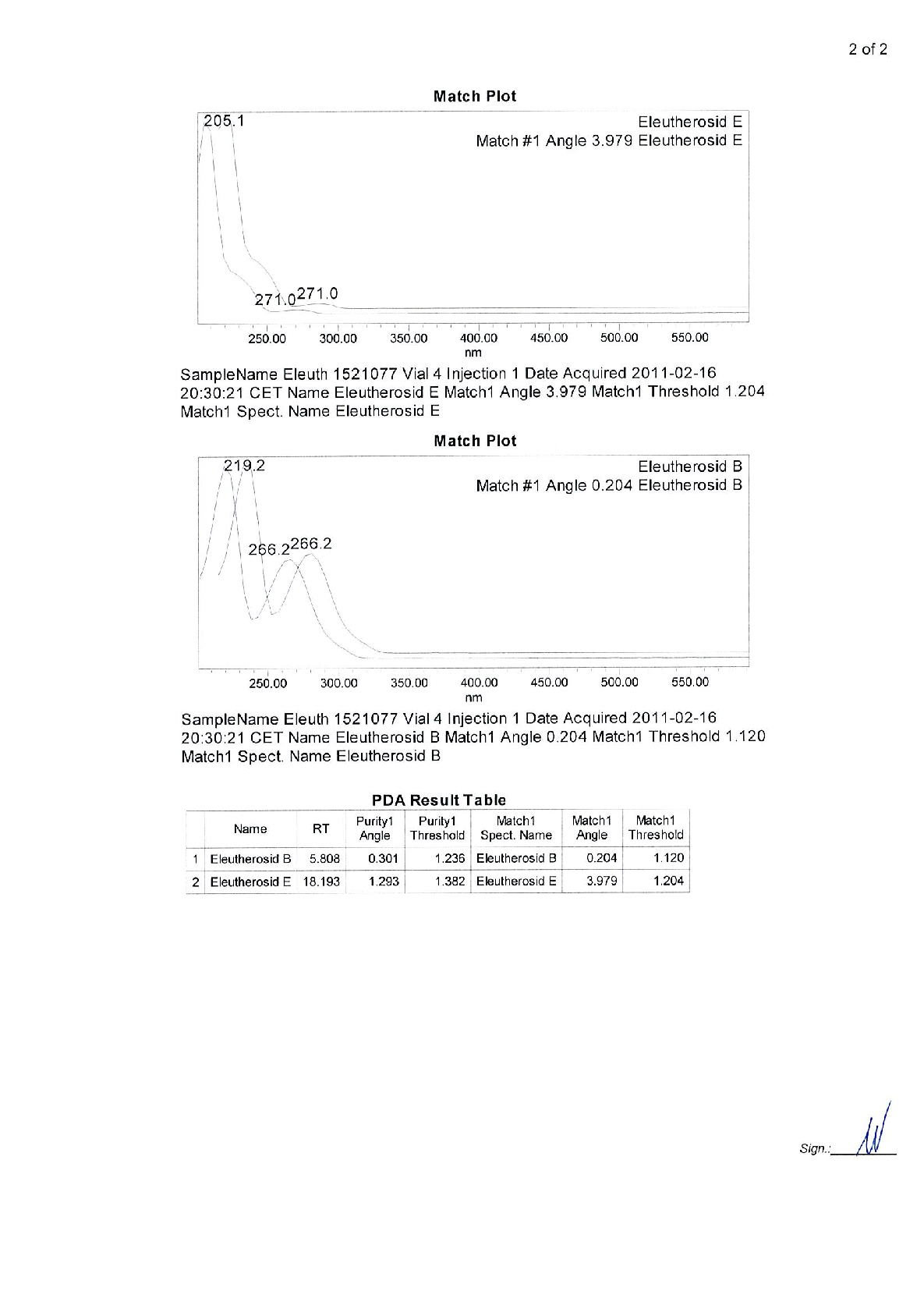
Figure 34
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart
Figure 35
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 36
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 37
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chart
Figure 38
Measured parameters from a study evaluating early intervention with Kan Jang® to treat upper, contributing to the overall assessment of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 39
Graphical representation of outcomes in a study of early intervention with Kan Jang® to treat upper, highlighting trends observed across experimental conditions.
chart
Figure 40
Quantitative data from a study on early intervention with Kan Jang® to treat upper, presenting measured outcomes relevant to the investigation of BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 41
Experimental results examining early intervention with Kan Jang® to treat upper, with data points illustrating key findings related to BACKGROUND AND AIM: Andrographis paniculata and Eleutherococcus senticosus preparations and their fixed combination, .
chart
Figure 42
Statistical analysis from research investigating early intervention with Kan Jang® to treat upper, comparing treatment groups and control conditions.
chartTables
Table 1
Table 2
Used In Evidence Reviews
Similar Papers
Planta medica · 2004
Andrographis paniculata in the treatment of upper respiratory tract infections: a systematic review of safety and efficacy.
Alternative medicine review : a journal of clinical therapeutic · 2007
Colds and influenza: a review of diagnosis and conventional, botanical, and nutritional considerations.
Cell death & disease · 2019
Inhibition of AIM2 inflammasome-mediated pyroptosis by Andrographolide contributes to amelioration of radiation-induced lung inflammation and fibrosis.
Journal of clinical pharmacy and therapeutics · 2004
Andrographis paniculata in the symptomatic treatment of uncomplicated upper respiratory tract infection: systematic review of randomized controlled trials.
Phytomedicine : international journal of phytotherapy and phytopharmacology · 2010
A randomized double blind placebo controlled clinical evaluation of extract of Andrographis paniculata (KalmCold) in patients with uncomplicated upper respiratory tract infection.
Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie · 2019