Modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products.
Study Design
- Çalışma Türü
- Review
- Popülasyon
- None
- Müdahale
- Modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products. None
- Karşılaştırıcı
- None
- Birincil Sonuç
- None
- Etki Yönü
- Mixed
- Yanlılık Riski
- Unclear
Abstract
Aging is a multifactorial process driven by accumulating cellular damage. Ferroptosis-an iron-dependent, lipid peroxidation-mediated form of cell death-has emerged as a critical contributor to age-related tissue degeneration. This review synthesizes current evidence linking ferroptosis to key aging hallmarks, including oxidative stress, chronic inflammation, mitochondrial dysfunction, and dysregulated iron metabolism. Central to these interactions is the age-associated decline in antioxidant defenses (e.g., glutathione, glutathione peroxidase 4 [GPx4]) and paradoxical iron dynamics, where systemic deficiency coexists with intracellular overload, promoting reactive oxygen species (ROS) generation via the Fenton reaction. Natural products such as resveratrol, curcumin, and epigallocatechin gallate (EGCG) exhibit promising anti-ferroptotic effects through mechanisms including iron chelation, ROS scavenging, and upregulation of endogenous antioxidants. Preclinical and clinical studies indicate their potential in reducing lipid peroxidation and enhancing cellular resilience in aging contexts. However, challenges such as poor bioavailability and tissue-specific iron dysregulation remain. This review explores how combinatorial approaches-targeting multiple ferroptosis pathways-may offer synergistic therapeutic benefits. Collectively, ferroptosis inhibition emerges as a promising strategy to mitigate age-associated tissue damage and promote healthy aging.
Kısaca
This review synthesizes current evidence linking ferroptosis to key aging hallmarks, including oxidative stress, chronic inflammation, mitochondrial dysfunction, mitochondrial dysfunction, and dysregulated iron metabolism to emerge as a promising strategy to mitigate age‐associated tissue damage and promote healthy aging.
Full Text
Review Article Modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products
Sherif Hamidu ,1 Seth Kwabena Amponsah ,2 Abigail Aning,1 Latif Adams,3 Justice Kumi ,1 Eunice Ampem-Danso ,1 Fatima Hamidu,4 Mustapha Abdul Mumin Mohammed,5 Gabriel Tettey Ador,6 and Sanjida Khatun7
- 1Department of Clinical Pathology, Noguchi Memorial Institute for Medical Research, College of Health Sciences, University of Ghana, Accra, Ghana
- 2Department of Medical Pharmacology, University of Ghana Medical School, College of Health Sciences, University of Ghana, Accra, Ghana
- 3Department of Microbiology and Immunology, School of Medical Sciences, College of Health and Allied Sciences, University of Cape Coast, Cape Coast, Ghana
4Faculty of Medicine, International University of Africa, Khartoum, Sudan
- 5Department of Internal Medicine, Central Hospital of Biel, Biel, Switzerland
- 6Department of Nutrition, Noguchi Memorial Institute for Medical Research, College of Health Sciences, University of Ghana, Accra, Ghana
7Biotechnology and Genetic Engineering, Faculty of Life Science, Mawlana Bhashani Science and Technology University, Santosh, Tangail, Bangladesh
Correspondence should be addressed to Sherif Hamidu; [email protected] and Seth Kwabena Amponsah; [email protected]
Received 8 April 2025; Revised 12 June 2025; Accepted 27 June 2025 Academic Editor: Udhaya Kumar Siva Kumar
Copyright © 2025 Sherif Hamidu et al. Journal of Aging Research published by John Wiley & Sons Ltd. Tis is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Aging is a multifactorial process driven by accumulating cellular damage. Ferroptosis—an iron-dependent, lipid peroxidationmediated form of cell death—has emerged as a critical contributor to age-related tissue degeneration. Tis review synthesizes current evidence linking ferroptosis to key aging hallmarks, including oxidative stress, chronic infammation, mitochondrial dysfunction, and dysregulated iron metabolism. Central to these interactions is the age-associated decline in antioxidant defenses (e.g., glutathione, glutathione peroxidase 4 [GPx4]) and paradoxical iron dynamics, where systemic defciency coexists with intracellular overload, promoting reactive oxygen species (ROS) generation via the Fenton reaction. Natural products such as resveratrol, curcumin, and epigallocatechin gallate (EGCG) exhibit promising anti-ferroptotic efects through mechanisms including iron chelation, ROS scavenging, and upregulation of endogenous antioxidants. Preclinical and clinical studies indicate their potential in reducing lipid peroxidation and enhancing cellular resilience in aging contexts. However, challenges such as poor bioavailability and tissue-specifc iron dysregulation remain. Tis review explores how combinatorial approaches—targeting multiple ferroptosis pathways—may ofer synergistic therapeutic benefts. Collectively, ferroptosis inhibition emerges as a promising strategy to mitigate age-associated tissue damage and promote healthy aging.
Keywords: aging; ferroptosis; iron metabolism; lipid peroxidation; natural products; oxidative stress
1. Introduction
Aging is a multifaceted biological process characterized by progressive accumulation of molecular and cellular damage. Te hallmarks of aging include dysbiosis and altered intercellular communication, with downstream consequences such as mitochondrial dysfunction, chronic infammation, and loss of proteostasis (Figure 1) [1]. Among these hallmarks, emerging evidence highlights ferroptosis—an iron-dependent regulated cell death driven by lipid peroxidation—as a critical yet underappreciated contributor to age-related tissue degeneration [2, 3]. Ferroptosis intersects with multiple aging pathways, including mitochondrial dysfunction (via redox imbalance), deregulated nutrient sensing (e.g., impaired glutathione synthesis), and epigenetic alterations (e.g., nuclear factor erythroid 2-related factor 2 [Nrf2] signaling suppression) [4–6]. Natural products, such as polyphenols, favonoids, and terpenoids, have been found to possess anti-aging properties. Tese natural products exhibit dual activity: ameliorate canonical aging hallmarks and suppress ferroptosis by scavenging lipid radicals, chelating iron, or upregulating antioxidant defenses [7, 8].
Indeed, the nexus between ferroptosis, aging hallmarks and natural products needs to be clearly aligned. For instance, an active compound from turmeric (curcumin) known to mitigate chronic infammation and telomere attrition also inhibits ferroptosis by activating the Nrf2-glutathione peroxidase 4 (GPX4) axis [3, 8, 9]. Similarly, resveratrol, a sirtuin-activating molecule that enhances proteostasis and autophagy, concurrently blocks ferroptotic death by modulating iron metabolism [10, 11]. Tis overlap suggests that some natural products with anti-aging properties exert their efects, in part, through ferroptosis inhibition. Tis may be a unifed strategy to target both the causes and consequences of aging.
In this review, we explore natural products and their ability to cause ferroptosis suppression; proposing the therapeutic utility of this in age-related diseases. By clearly defning the hallmarks of aging with ferroptosis pathways, we aim to unravel how nature-derived compounds could delay aging and resolve the ferroptotic “tipping point” that accelerates cellular collapse.
Tis review article specifcally aligns with the scope of the Journal of Aging Research by critically evaluating the intricate relationship between ferroptosis and the aging process, and by exploring the therapeutic potential of natural products in modulating this cellular death pathway. Our comprehensive summary aims to provide novel insights into the mechanistic underpinnings of age-related diseases and propose translational strategies for healthy aging, thereby contributing to the journal’s mission of advancing knowledge in gerontology and geriatric medicine.
1.1. Aging: An Unavoidable Biological Process. As one ages, there is gradual decline in the functional capacity of cells, tissues, and organ systems, thereby heightening the risk of frailty, disease, and eventual death [12, 13]. Aging arises from a complex network of intrinsic mechanisms, including disruptions in the balance between pro-oxidant and antioxidant
forces [14], shifts in anabolic and catabolic processes [15, 16], disturbances in energy metabolism [17], and the concurrent activation of various immune responses [18]. Collectively, these factors foster a persistent, low-level infammatory state that leads to immune senescence, setting of a self-reinforcing cycle that accelerates further deterioration [19]. Recognized hallmarks of aging across species include genomic instability, telomere shortening, epigenetic changes, loss of proteostasis, impaired nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell depletion, and altered cell-to-cell communication [20–22]. Notably, many of these hallmarks such as mitochondrial dysfunction and redox imbalance converge with ferroptosis, an iron-dependent cell death pathway driven by lipid peroxidation, which exacerbates age-related decline [23, 24]. However, the intricate process of aging remains only partially understood, with many aspects yet to be unraveled.
- 1.2. Evolving Mechanisms of Aging. Originally described by Denham Harman in the 1950s, the free radical theory posits that aging results from the cumulative oxidative damage inficted by reactive oxygen species (ROS) generated during normal cellular metabolism, which progressively shortens lifespan [25, 26]. Alongside this widely recognized concept, new ideas propose that the inherent imperfections of biological systems also contribute to aging [25]. Cells continuously get damaged due to intrinsic heterogeneity and imperfect fdelity of biological processes, eventually leading to senescence [27, 28]. Te rate at which these damages accrue is contingent upon the efciency of metabolic and genetic repair systems. Recent fndings indicate that ROS alone cannot fully account for the aging process [29]. Ferroptosis, a form of regulated necrosis driven by iron overload and lipid peroxidation, has emerged as another important contributor to aging, particularly in tissues prone to redox imbalance (e.g., brain and liver) [3, 4, 30]. It is, therefore, relevant to explore other pathways of cellular damage that play roles in initiating, sustaining, and advancing aging [31].
- 1.3. Chronic Infammation, Aging, and Disease. Te term “infamm-aging,” introduced in 2000 [32], characterizes aging as a process accompanied by a continuous, low-grade, systemic, and unresolved infammatory state that gradually increases pro-infammatory markers [33]. Tis persistent infammation is believed to be a key determinant of both the rate of aging and overall lifespan. Several studies have implicated such sustained, mild infammation as a signifcant risk factor for age-related conditions including atherosclerosis, arthritis, cancer, diabetes, osteoporosis, dementia, vascular disorders, obesity, and metabolic syndrome [33, 34]. Aging is further associated with an imbalance in redox homeostasis, where the chronic upregulation of proinfammatory mediators (e.g., TNF-α, IL-1β, IL-6, COX-2, iNOS) and the activation of pathways like NF-κB occur alongside diminished antioxidant defenses [35, 36]. Tis infammatory milieu not only accelerates cellular senescence but also primes cells for ferroptosis by depleting glutathione (a key antioxidant) and increasing labile iron pools [37]. Te precise cause-and-efect relationship between chronic
Hallmarks of aging
Telomere attrition
Dysbiosis
Chronic inflammation
Genome instability & mutation
Altered intercellular communication
Epigenetic alterations
Stem cell exhaustion
Aging Loss of proteostasis
Disabled macroautophagy
Cellular senescence
Mitochondrial dysfunction
Deregulated nutrient sensing
Primary hallmarks Antagonistic factors Integrative hallmarks
- Figure 1: Interconnected hallmarks of aging. Tis schematic diagram depicts the multifaceted processes contributing to aging, encompassing key hallmarks such as dysbiosis, chronic infammation, mitochondrial dysfunction, and disrupted proteostasis. It emphasizes how imbalances in nutrient sensing, genomic integrity, epigenetic regulation, and autophagy converge to drive cellular senescence, stem cell depletion, and systemic decline. Antagonistic factors (e.g., chronic infammation) and integrative features (e.g., altered intercellular communication) further intensify ageassociated dysfunction through a self-perpetuating cycle. Highlighted pathways—including mitochondrial dysfunction and redox imbalance—intersect with ferroptosis, ofering mechanistic insights into the aging process and potential therapeutic targets.
- infammation and age-associated diseases remains unclear; however, current evidence suggests a vicious cycle of enhanced frailty, accelerated aging, and premature death (Figure 2).
- 1.4. Iron Metabolism and Aging. Recently, there is a lot of attention on iron dysregulation and aging. Indeed, imbalances in iron metabolism are common in older individuals. For instance, iron defciency anemia, marked by low serum ferritin and reducedsystemiciron, isprevalent among theelderly andis associated with adverse outcomes such as cardiovascular disease, increased falls and fractures, cognitive decline, diminished quality of life, and heightened mortality risk [38–40]. Beyond poor nutrition and the use of certain medications, elevated circulating hepcidin levels, often stemming from chronic infammation, may also contribute to systemic iron depletion [41]. Nonetheless, further research is needed to clarify the complex interactions between aging, iron status, and the regulation of hepcidin (a key regulator for the entry of iron into the circulation of mammals) [38, 39]. Additionally, aging is accompaniedby increasedintracellular iron accumulation, which, due to redox imbalances, can trigger ferroptosis—a form of cell death that exacerbates age-related functional decline and mortality [40]. Tis paradox highlights the potential of natural products (e.g., curcumin, quercetin) to modulate iron homeostasis and inhibit ferroptosis by chelating excess iron or
upregulating antioxidant defenses [42, 43]. Te paradox of low systemic iron combined with elevated intracellular iron might be driven by hepcidin upregulation in response to chronic infammation [42]. Tus, targeting hepcidin and its regulatory pathways could represent a promising strategy to mitigate agerelated decline and associated diseases.
1.5. Age-Related Iron Dysregulation and Ferroptosis. Eukaryotic cells rely on iron to support essential biological functions including energy production, deoxyribonucleic acid (DNA) synthesis, replication, and detoxifcation. Although iron is indispensable for growth and development, its levels are strictly controlled by a network of transporters, storage proteins, and regulators to prevent both defciency and toxicity [44, 45]. Despite these rigorous homeostatic mechanisms, the body lacks an efcient excretion system for iron [45]. Iron loss occurs primarily through bleeding or the natural shedding of cells, resulting ina minimal daily loss (about 1mg) compared to approximately 4g stored in the body, while the duodenum absorbs roughly 1mg of iron per day [44, 46, 47]. Insufcient iron during development can impair key physiological processes,whileexcessiveironretentioninadulthoodislinkedwith acceleratedaging.Contributingfactorstoironoverloadinaging include: (i) reduced metabolic demand for iron by cofactordependent enzymes; (ii) decreased hemoglobin levels, which account for about 60% of total body iron; and (iii) the relative
Aging
Pathological Antioxidant cycle
ROS
Ferroptosis Ferroptosis
- Figure 2: Pathological cycle in aging. Schematic diagram illustrating the vicious cycle of ferroptosis during aging and its contribution to chronic diseases. Excessive reactive oxygen species and iron-dependent lipid peroxidation trigger ferroptosis, driving pathological processes that accelerate age-related organ damage. Antioxidant interventions can disrupt this cycle, highlighting a potential therapeutic strategy to mitigate chronic disease progression in aging populations.
iron overload observed in post-menopausal women. Over a lifetime, the buildup of iron in somatic tissues can disrupt cellularfunctions,trigger celldeath,and promoteaging[44,47]. Compounds found in natural products such as polyphenols (e.g., resveratrol) and favonoids (e.g., epigallocatechin gallate [EGCG]) may counteract this iron buildup by restoring iron homeostasis and suppressing ferroptosis, thereby delaying agerelated pathologies [43]. It is hypothesized that this ageassociated iron imbalance may be associated with ferroptosis, a unique, iron-dependent form of regulated cell death.
- 1.6. Ferroptosis: A Distinct Mode of Cell Death. Traditionally, cell death can be categorized into necrosis, apoptosis, and autophagy [48]. However, other nonapoptotic mechanisms exist, one of which is ferroptosis. Defned as an iron-dependent form of regulated necrosis, ferroptosis is triggered by extensive lipid peroxidation that damages cellular membranes [31]. Its involvement in conditions such as cardiovascular diseases, cancers, and neurological disorders is well documented [31, 37]. As a result, ferroptosis inhibitors, including natural products like ferrostatin-1 and liproxstatin-1, have been discovered [49]. Te term “ferroptosis” was introduced in 2012 following the discovery of small molecules that selectively inhibited the growth of RAS-mutant cancer cells [50]. Early hypotheses regarding ferroptosis emerged from observations in nutrient-deprived cancer cells [51] and from studies on
“oxytosis”—a phenomenon where neurons die due to glutamate toxicity coupled with inhibition of the amino acid antiporter SLC7A11/xCT/system xc− [52, 53].
2. Types of Cell Death: From Apoptosis to Ferroptosis and Beyond
Cell death can occur through several pathways (Figure 3), each defned by unique morphological and biochemical signatures [48]. Te principal modes include:
- • Apoptosis:
- • Necrosis:
- • Regulated Necrosis (Necroptosis):
Although similar to necrosis in terms of outcome, necroptosis is a programmed process mediated by
Cell death
Cell death (healthy)
Programmed cell death
Unprogrammed cell death
Necrosis
Necroptosis Pyroptosis
Apoptosis
Ferroptosis Autophagy
- Figure 3: Classifcation of cell death mechanisms. Tis fgure classifes various forms of cell death, including apoptosis, necrosis, pyroptosis, autophagy-associated death, necroptosis, and ferroptosis. Apoptosis is a noninfammatory programmed cell death; necrosis is uncontrolled and infammatory. Pyroptosis involves infammatory caspases, while ferroptosis is characterized by iron accumulation and lipid peroxidation. Each pathway is regulated by distinct molecular mechanisms.
specifc signaling molecules such as RIPK1, RIPK3, and MLKL. Tis pathway is often activated when apoptotic machinery is inhibited, and this is implicated in various infammatory conditions.
- • Autophagy-Associated Cell Death:
- • Pyroptosis:
- • Ferroptosis:
Distinct from the above, ferroptosis is an irondependent, regulated form of cell death triggered by the accumulation of lipid peroxides. Unlike apoptosis or necroptosis, ferroptosis is defned by its reliance on iron metabolism, failure of the glutathione-dependent antioxidant defense (especially GPx4), and extensive membrane damage. Emerging studies highlight natural compounds such as curcumin and resveratrol as dual-acting agents that inhibit ferroptosis by both
chelating iron and enhancing GPx4 activity, ofering a strategic avenue to decelerate aging [24, 31]. Its emergence as a signifcant cell death mechanism has spurred intense research into its roles in various diseases, including neurodegeneration, cancer, and—importantly—aging.
2.1. Mechanism of Ferroptosis. Ferroptosis is a distinct, iron-dependent form of regulated cell death that difers from apoptosis and necroptosis, occurring independently of apoptotic (e.g., BAX, BAK, and caspases) and necroptotic (e.g., MLKL, RIPK1, and RIPK3) efectors [48, 50]. While ferroptosis plays a tumor-suppressive role by eliminating malignant cells, it is also implicated in various diseases, where it contributes to pathogenic mechanisms that were previously unexplained.
Te progression of ferroptosis involves four key steps (Figure 4):
- 1. Cysteine Uptake Inhibition: Te system Xc antiporter, which imports cystine (Cys) in exchange for glutamate (Glu), is inhibited. Tis reduces intracellular cysteine levels, limiting the synthesis of glutathione (GSH), a crucial antioxidant.
- 2. Glutathione and GPX4 Depletion: Reduced GSH synthesis leads to decreased activity of GPX4, an enzyme that normally prevents lipid peroxidation.
Ferroptosis Transferrin (Tf) with signaling pathway
2 iron molecules Tf
Tf
Cys Glu Gln
Transferrin receptor Glutamate-cystine antiporter (Xc)
SLC1A5
Cys Glu
Endosomal uptake
Gln
GLS
Fe3+
GCL GSS
STEAP3
+ Gly
Fe2+
DMT1 Ferritin storage
GSH
PUFA-OH GPX4
PUFA-OOH
Fenton reaction
Lipid
Lipoxygenases
Lipid peroxidation
P450 oxidoreductase
Ferroptosis
- Figure 4: Ferroptosis signaling pathway. Tis schematic diagram outlines the molecular cascade of ferroptosis. Cystine uptake via the system Xc⁻ antiporter is inhibited, reduced GSH synthesis. Tis impairs GPx4, an enzyme that detoxifes lipid peroxides. Te resulting lipid peroxidation is amplifed by iron (Fe2⁺)-driven Fenton reactions, producing ROS. Tf- mediated iron uptake and its reduction via STEAP3 and DMT1 contribute to intracellular iron overload, further exacerbating ferroptosis. Abbreviations: GSH-glutathione; GPx4-glutathione peroxidase 4; ROS-reactive oxygen species; Tf-transferrin; DMT1-divalent metal transporter 1; STEAP3-six-transmembrane epithelial antigen of prostate 3.
- 3. Excessive Lipid ROS Accumulation: With GPX4 inactivated, polyunsaturated fatty acids (PUFAs) undergo peroxidation, forming toxic lipid peroxides (PUFA-OOH), which accumulate. Tis step is driven by enzymes such as lipoxygenases, cytochrome P450 oxidoreductases, and non-enzymatic Fenton chemistry, where Fe2+ reacts with hydrogen peroxide (H2O2) to generate ROS.
- 4. Iron Overload and ROS-Driven Peroxidation: Increased intracellular iron, facilitated by transferrin (Tf) uptake and its conversion from Fe3+ to Fe2+ via STEAP3 and DMT1, fuels lipid peroxidation through the Fenton reaction, exacerbating oxidative damage and ultimately leading to ferroptosis.
A defning ultrastructural feature of ferroptosis, observed via transmission electron microscopy, includes shrunken mitochondria with increased membrane density and reduced cristae structure, signifying impaired cellular energetics [53].
Beyond the four key steps of ferroptosis, the integral role of mitochondria, their function, damage, and repair mechanisms, are increasingly recognized as critical nodes
in this unique form of cell death. A defning ultrastructural feature of ferroptosis, observed via transmission electron microscopy, includes shrunken mitochondria with increased membrane density and reduced cristae structure, signifying impaired cellular energetics [53]. Mitochondrial dysfunction, characterized by excessive mitochondrial ROS production, disruption of mitochondrial membrane potential, and impaired bioenergetics, directly fuels the lipid peroxidation cascades central to ferroptosis. Tis mitochondrial involvement is particularly relevant in aging, where mitochondrial dysfunction is a well-established hallmark, contributing to cellular senescence and tissue degeneration [7, 53]. Furthermore, compromised mitochondrial quality control mechanisms, such as dysregulated mitophagy (the selective removal of damaged mitochondria), can exacerbate ferroptosis susceptibility in aging cells. Terefore, understanding and targeting mitochondrial integrity and function represents a crucial avenue for modulating ferroptosis in age-related contexts. Despite extensive research, critical aspects of ferroptosis regulation remain unresolved, highlighting its potential therapeutic implications in both oncology and degenerative diseases.
Natural antioxidants such as silymarin and α-lipoic acid may counteract ferroptosis by replenishing glutathione levels or scavenging lipid radicals. Emerging evidence suggests that ferroptosis can spread paracrine-like signals through aldehydes such as 4-hydroxynonenal (4-HNE) and malondialdehyde (MDA), which react with cellular macromolecules at sites distant from the initial damage [54].
mitigate ferroptosis in aging focus on Nrf2 signaling pathway (Figure 6).
- 2.3.1. Resveratrol: Activating Nrf2 to Restore Redox Balance. Resveratrol, a polyphenol prevalent in grapes and red wine, exerts its antioxidant efects by activating the Nrf2 pathway [69]. Tis activation leads to increased synthesis of GSH and upregulation of HO-1, which together help neutralize lipid peroxides and chelate labile iron. In aged neuronal models, resveratrol was found to reduce lipid peroxidation by approximately 40% and enhanced GPx4 activity, thereby protecting cells from ferroptosis [70–73]. Additionally, there is compelling evidence that shows that resveratrol extended the lifespan of C. elegans by suppressing mitochondrial ROS and mitigating iron accumulation, underscoring its dual function as both an iron modulator and Nrf2 activator: a combination that renders it a potent inhibitor of ferroptosis in aging tissues [74–76].
- 2.3.2. Curcumin: Chelating Iron and Boosting GPx4. Curcumin, a bioactive compound derived from turmeric, functions as both an efective iron chelator and a modulator of antioxidant defenses. Its unique β-diketone structure allows it to bind excess iron, thereby preventing the Fenton reaction and the consequent production of ROS [77]. Additionally, curcumin upregulates GPx4, the key enzyme responsible for detoxifying lipid hydroperoxides. In aged mice, curcumin supplementation led to a 30% increase in GPx4 expression and a reduction in hippocampal markers of ferroptosis, which was associated with improved cognitive performance [78]. Furthermore, a clinical trial in 2022 reported that elderly subjects taking curcumin exhibited a 25% decrease in serum MDA, a biomarker of lipid peroxidation, further supporting its protective role [79, 80]. Tese fndings underscore curcumin’s dual action as an iron chelator and GPx4 enhancer; highlighting its potential to combat agerelated neurodegeneration.
- 2.3.3. EGCG: Targeting Lipid Peroxidation. EGCG, a catechin in green tea, exhibits potent antioxidant properties by scavenging lipid radicals and inhibiting pro-oxidant enzymes, including NADPH oxidase [81]. Additionally, EGCG has been shown to upregulate antioxidant systems, such as superoxide dismutase, catalase, and GSH, thereby enhancing the body’s defense against oxidative stress [81]. In aged rat models, EGCG administration resulted in a signifcant reduction of lipid peroxidation markers and preservation of mitochondrial integrity [82]. Furthermore, in rats, EGCG has demonstrated neuroprotective efects by inhibiting neuronal cell death and improving cerebral function following traumatic brain injury [82]. Tese fndings highlight EGCG’s multifaceted role in directly inhibiting lipid peroxidation and modulating iron-related pathways.
- 2.3.4. Sulforaphane: Amplifying Endogenous Antioxidants. Sulforaphane, a bioactive compound abundant in broccoli sprouts, enhances the body’s antioxidant defenses by
- 2.2. Oxidative Stress, Ferroptosis, and Aging. Elevated iron levels can precipitate ferroptosis by catalyzing ROS production through the Fenton reaction. Tis suggests that both increased iron uptake and excessive iron storage may contribute to ferroptotic cell death [55, 56]. Specifcally, the divalent ferrous ion (Fe2+) reacts with hydrogen peroxide
- 2.3. Natural Compounds Targeting Ferroptosis. Natural products and medicinal plants have been used to treat various diseases including infections and cancers [63–65]. As antioxidant defenses wane with aging [7], cells become increasingly vulnerable to ferroptotic damage, accelerating tissue degeneration and diseases like neurodegeneration and cardiovascular disorders. Natural products, with pleiotropic antioxidants [66, 67], anti-infammatory and iron-chelating properties [68] ofer promise in decreasing ferroptotic damage. Reports show that key compounds (Figure 5) that
HO OH
OH
HO
O
O
OH
HO
HO OH
O
HO
O
OH
O
O O
HO
OH
OH
I II III
O
OH
HO
O
S
S
HO
H3C N
O
OH
HO
IV V
OH
HO
OH
O
HO
O
VI VII VIII
CH2
O
OH
CH2
OH OH
O
OH
OH
IX X
OH HO HO
O
CH3
HO
CH3 CH
CH3
H C
CH
O
H3C
O
OH
O
O
CH3 CH3 CH3
H3C
OH O
O
OH
XI XII
OH
OH
O
O
O
OH
O
H3C CH3
HO
O
O OH
OH
OH
HO
HO
XIII XIV XV
CH
O
O
H3C
CH3
HO O
H3C
OH
HO
OH
O
CH3 H C
CH3
O
CH
H3C
OH
O O
OH
HO
XVI XVII XVIII
OH
OH HO O
HO
CH3
HO
O
OH
OH
CH3
O H3C
HO
O
HO
H3C
OH
O
HO
XIX XX
- Figure 5: Chemical structures of natural compounds modulating ferroptosis. Images generated using ChemSketch illustrate the molecular structures of 20 key natural compounds known to inhibit ferroptosis by targeting iron metabolism, lipid peroxidation, and antioxidant pathways. Te compounds are listed as follows: (I) resveratrol; (II) curcumin; (III) EGCG; (IV) sulforaphane; (V) quercetin; (VI) coenzyme Q10; (VII) lycopene; (VIII) fsetin; (IX) baicalein; (X) honokiol; (XI) silymarin; (XII) astaxanthin; (XIII) apigenin; (XIV) luteolin; (XV) gingerol; (XVI) ursolic acid; (XVII) ellagic acid; (XVIII) pterostilbene; (XIX) carnosol; and (XX) theafavins. Tese compounds exert pleiotropic efects such as activating Nrf2, enhancing GPx4, and scavenging ROS.” Abbreviations: EGCG-epigallocatechin gallate; GPx4glutathione peroxidase 4; ROS-reactive oxygen species; Nrf2-nuclear factor erythroid 2-related factor 2.
Expression of antioxidant proteins (GSH AND HO-1) BY Nrf2
Constitutive conditions Nrf2 activation
Resveratrol Sulforaphane Gingerol
Transferrin (Tf) with 2 iron molecules
RBX1 Cul3
Actin
Keap1
Keap1
Keap1
Keap1
Ubiquitination
Tf
Nrf2
Nrf2 release
Tf
Cys Glu Gln
Nrf2
Nrf2
Nrf2
Nrf2
Transferrin receptor Glutamate-cystine antiporter (Xc)
SLC1A5
Nrf2 nuclear translocation
Nrf2
Endosomal uptake
Cys Glu
Gln
GLS
Fe3+
Nucleus
GCL GSS
+ Gly
STEAP3
Antioxidant proteins expression
Fe2+
Stimulates
Ferritin storage
Nrf2
sMaf
Nrf2
GSH
DMT1
ARE Cytoplasm Degraded Nrf2
PUFA-OH GPX4
PUFA-OOH
Ferroptosis
Fenton reaction
Lipid
Lipoxygenases
Lipid peroxidation
P450 oxidoreductase
Ferroptosis
Aging
Natural products
- • Curcumin
- • Silymarin
Increases the expression of GPx4
- Figure 6: Activation of Nrf2 and its role in antioxidant defense against ferroptosis. Tis schematic diagram illustrates the regulation of Nrf2 under constitutive conditions (left panel) and upon activation (middle and right panels). Under basal conditions, Nrf2 is ubiquitinated and degraded via the Keap1-Cul3-RBX1 complex. Upon activation by compounds such as resveratrol, sulforaphane, and gingerol, Nrf2 is released from Keap1, translocates to the nucleus, and binds to ARE, leading to the expression of antioxidant proteins. Te right panel highlights the role of Nrf2 in regulating GSH and HO-1, which protect against lipid peroxidation and ferroptosis. Additionally, curcumin and silymarin enhance GPx4 expression, a key enzyme mitigating lipid peroxidation. Abbreviations: Nrf2-nuclear factor erythroid 2-related factor 2; Keap1-Kelch-like ECH-associated protein 1; ARE-antioxidant response element; GSH-glutathione; HO-1-heme oxygenase-1; GPx4-glutathione peroxidase 4.
activating the Nrf2 pathway. Tis activation leads to increased synthesis of GSH, a crucial endogenous antioxidant, and upregulation of various cytoprotective proteins [83]. A study demonstrated that sulforaphane induces the translocation of Nrf2 into the nucleus, increasing the expression of γ-glutamylcysteine synthetase (γ-GCS), the rate-limiting enzyme in GSH synthesis, thereby raising intracellular GSH levels [84]. Tese fndings highlight sulforaphane’s potential in enhancing antioxidant capacity, ofering protection against oxidative stress and age-related cellular decline.
- 2.3.5. Quercetin: Iron Sequestration and Sirtuin 1 (SIRT1) Activation. Quercetin, a favonoid abundantly found in apples and onions, not only chelates free iron to reduce its intracellular levels but also activates SIRT1, a key longevityassociated deacetylase that bolsters cellular stress resistance. In studies using senescent fbroblasts, quercetin treatment reduced intracellular iron by approximately 25% and suppressed ACSL4, an enzyme critical for pro-ferroptotic PUFA synthesis, through a mechanism involving SIRT1 activation [85]. Moreover, a 2022 study demonstrated that quercetin improved cardiac function in aged mice by inhibiting ferroptosis, underscoring its dual role in both iron
sequestration and the epigenetic regulation of genes that drive ferroptotic cell death [86]. Together, these fndings highlight quercetin’s promising therapeutic potential in mitigating age-related tissue dysfunction through the combined actions of iron chelation and enhanced stress resistance via SIRT1.
2.3.6. CoQ10: Preserving Mitochondrial Resilience. CoQ10 is a lipid-soluble antioxidant integral to mitochondrial electron transport and cellular energy production. Beyond its role in adenosine triphosphate (ATP) synthesis, CoQ10 exhibits iron-chelating properties that mitigate oxidative stress, particularly within mitochondria. In models of iron overload-induced damage, CoQ10 administration was found to alleviate oxidative injury by chelating excess iron, thereby reducing ROS production and preserving mitochondrial integrity [87]. Additionally, CoQ10 infuences the activity of SIRT1, an NAD+ -dependent deacetylase associated with longevity and metabolic regulation. Studies indicate that CoQ10 defciency can compromise SIRT1 activity, suggesting that adequate CoQ10 levels are essential for optimal SIRT1 function [88]. Furthermore, a clinical trial demonstrated that supplementation with CoQ10 and
1: Mechanisms of natural compounds in inhibiting ferroptosis.Table
in ferroptosis Impact on agingReference
Alzheimer’s models. [111–113]
models. [100–103]
damage. [104–106]
delays sarcopenia. [108–110]
vascular infammation. [114–116]
markers in aged rats. [117–119]
senescence in aging models. [98–100]
aged mice. [92, 93]
potential in elderly subjects. [94, 95]
markers in elderly subjects. [96, 97]
preserves mitochondrial integrity. [90, 91]
function. [107]
C. elegansneurons; extends lifespan in. [69]
serum MDA by 25% in elderly subjects. [78]
in plasma GSH in elderly cohorts. [83]
uptake. Lowers 4-HNE by 35% in aged brains;
ferroptosis suppression). 50% increase in hepatic FSP1; 20% rise
membranes. 30% lower ROS in aged skeletal muscle;
production; upregulates FSP1. Protects aged endothelial cells; reduces
defenses. Reduces neuronal ferroptosis by 25% in
ferroptosis markers in oxidative stress
GPx4 and HO-1; chelates labile iron. Reduces lipid peroxidation by 40% in
protects against age-associated neural
GPx4 expression. 30% increase in GPx4 levels; reduces
antioxidant defenses. 25% decrease in ROS; delays cellular
incorporation. 20% reduction in cardiac ferroptosis
iron-induced oxidative stress. Improves neuronal survival; reduces
peroxides via favonolignans. Reduces liver ferroptosis by 35% in
enzyme). 25% reduction in intracellular iron;
from lipid peroxidation. 30% reduction in oxidative damage
improves mitochondrial membrane
imMDA proves cardiac function in
lipid peroxidation. 30% reduction in ROS generation;
electron transport to reduce ROS. 20% reduction in plasma MDA;
aged rodents; improves hepatic
SulforaphaneBroccoli sprouts Activates Nrf2, boosting GSH synthesis
SilymarinMilk thistle Enhances GPx4 activity; scavenges lipid
HonokiolMagnolia bark Suppresses iron-induced ROS; inhibits
(FSP1) expression (GPx4-independent
downregulate ACSL4 (pro-ferroptotic
and ferroptosis suppressing protein 1
QuercetinApples, onions Chelates free iron; activates SIRT1 to
LycopeneTomatoes Scavenges ROS; protects membranes
Scutellaria baicalensisBaicalein Inhibits lipid peroxidation; mitigates
FisetinStrawberries, apples Reduces intracellular ROS; enhances
ResveratrolGrapes, red wine Activates Nrf2 pathway, upregulates
AstaxanthinMicroalgae, seafood Neutralizes singlet oxygen and lipid
membranes; sustains mitochondrial
15-LOX, blocking PUFA oxidation;
enhance mitochondrial antioxidant
CurcuminTurmeric βBinds iron via-diketone groups, inhibits fenton reactions; enhances
downregulates TfR1 to limit iron
ApigeninParsley, chamomile Chelates iron; activates SIRT3 to
radicals; stabilizes mitochondrial
suppresses ACSL4-driven PUFA
Scavenges lipid radicals; inhibits
GingerolGinger Modulates Nrf2/ARE pathway;
LuteolinCelery, broccoli Inhibits NOX4-mediated ROS
Coenzyme Q10Dietary supplements Neutralizes lipid peroxides in
CompoundSource Mechanism of action
gallate (EGCG) Green tea
Epigallocatechin
T1: Continued.able
potential: pairing iron chelators (e.g., quercetin) with Nrf2 activators (e.g., sulforaphane) may enhance efcacy. Bioavailability challenges: Some compounds (e.g., curcumin, resveratrol) require formulation
Note:Key targets include iron chelation, Nrf2/GPx4 activation, and lipid peroxidation blockade. Mechanistic diversity: compounds target iron metabolism (e.g., quercetin, ellagic acid), lipid peroxidation (e.g.,
EGCG, astaxanthin), and antioxidant defense (e.g., sulforaphane, resveratrol). Clinical relevance: curcumin, CoQ10, and lycopene have human trial data; others are supported by preclinical models. Synergy
in ferroptosis Impact on agingReference
reduces serum creatinine by 20%. [131, 132]
reduces mitochondrial ROS. [120, 121]
brain lipid peroxidation. [125–127]
markers in aged joints. [128–130]
mice; reduces hepatic fbrosis. [122–124]
restriction. Extends lifespan in drosophila; reduces
oxidase-driven ROS production. Protects aged kidneys from ferroptosis;
autophagy. Improves muscle health in aged mice;
scavenging. 25% lower liver iron content in aged
arachidonic acid peroxidation. 30% reduction in infammatory
Ursolic acidApple peels, rosemary Activates AMPK to inhibit mTOR and
CarnosolRosemary Inhibits 5-LOX and COX-2; reduces
PterostilbeneBlueberries Activates SIRT1 to deacetylate and
reduce iron absorption; enhances
TeafavinsBlack tea Chelates iron; inhibits xanthine
peroxidation via direct radical
stabilize GPx4; mimics caloric
Ellagic acidPomegranate, berries Chelates iron; inhibits lipid
CompoundSource Mechanism of action
improvements for optimal delivery.
selenium led to increased SIRT1 concentrations in elderly subjects, highlighting its potential in modulating pathways linked to aging and cellular stress responses [89]. Collectively, these fndings underscore CoQ10’s multifaceted role in preserving mitochondrial resilience through iron chelation and SIRT1 activation.
- 2.4. Natural Products and Potential Antiaging Properties. Natural compounds such as resveratrol, sulforaphane, curcumin, quercetin, EGCG, and CoQ10 have demonstrated the ability to modulate ferroptosis pathways (Table 1), offering potential therapeutic avenues for mitigating agingrelated cellular damage. Tese compounds target critical aspects of ferroptosis, including iron metabolism, lipid peroxidation, and antioxidant defense mechanisms. For instance, resveratrol and sulforaphane enhance the Nrf2GPx4 signaling pathway, counteracting age-associated GSH depletion [133]. Curcumin and quercetin exhibit ironchelating properties, thereby reducing Fenton chemistryinduced oxidative stress. EGCG and CoQ10 stabilize cellular membranes against lipid peroxidation, preserving mitochondrial integrity. Combining resveratrol with quercetin may synergistically enhance their efcacy. Ongoing clinical trials, like the SPRINTT project, are investigating multicomponent interventions, including physical activity and nutritional counseling, to prevent mobility disability in frail older adults [134]. Tese studies aim to validate the therapeutic potential of such natural compounds in aging populations.
3. Discussion
Te inexorable progression of aging occurs as a result of molecular and cellular dysregulation, among which ferroptosis has emerged as playing a role in redox imbalance, chronic infammation, and tissue degeneration [4]. Ferroptosis has been found as both a consequence and accelerator of aging, partly explaining the cumulative cellular damage of age-related pathologies. Here, we contextualize these fndings within aging biology and highlight the therapeutic potential of natural compounds in modulating ferroptotic pathways.
Aging is marked by the progressive erosion of homeostatic systems, including redox regulation, proteostasis, and nutrient sensing. Central to this decline is the dysregulation of iron metabolism, which creates a permissive environment for ferroptosis. Elevated intracellular iron, a hallmark of aging, catalyzes the Fenton reaction, generating hydroxyl radicals that propagate lipid peroxidation in membranes rich in PUFAs [135, 136]. Te age-associated decline in GSH and GPx4, critical guardians against lipid peroxide accumulation, renders cells vulnerable to ferroptotic death [137]. Tis vulnerability is exacerbated by chronic infammation (“infamm-aging”), which does not only deplete antioxidant reserves but also upregulates hepcidin, trapping iron within cells and further fueling oxidative damage [138, 139]. Our review aligns with recent studies implicating ferroptosis in neurodegenerative diseases [140], cardiovascular
dysfunction [141], and sarcopenia [135], suggesting its broad role in age-related morbidity.
Compounds from natural products, with their pleiotropic mechanisms, represent a promising strategy to disrupt ferroptosis and its contribution to aging (Table 1). For instance:
- • Resveratrol activates the Nrf2 pathway, upregulating GPx4 and HO-1 to neutralize lipid peroxides and chelate labile iron [9]. In preclinical models, resveratrol reduced hippocampal lipid peroxidation by 40%, which led to improved cognitive function in aged mice [69].
- • Curcumin directly binds iron via its β-diketone structure, inhibiting Fenton chemistry, while enhancing GPx4 expression [78]. A 2022 clinical trial demonstrated a 25% reduction in serum MDA in elderly subjects supplemented with curcumin [146].
- • EGCG is known to scavenge lipid radicals and inhibit 15-lipoxygenase (15-LOX), blocking PUFA oxidation [90, 91]. In aged rat brains, EGCG lowered 4-HNE by 35%, preserving mitochondrial integrity [90, 91].
Tese compounds exemplify a multitarget approach, addressing iron homeostasis, antioxidant defense, and infammatory signaling simultaneously. Such synergy is critical in aging, where single-target therapies often fail to address the multifactorial nature of decline. While preclinical data are compelling, clinical validation remains limited. For example, sulforaphane, an Nrf2 activator, has been shown to increase Nrf2 transcription, activation, nuclear translocation, DNA-binding, and antioxidant gene expression in epithelial cells isolated from elderly humans [147]. Similarly, CoQ10 supplementation has been observed to reduce lipid peroxidation levels in humans [148]. A key challenge lies in the paradoxical iron dynamics of aging: systemic iron defciency (e.g., anemia) coexisting with intracellular iron overload, complicating therapeutic iron modulation. Natural chelators like quercetin may ofer a balanced approach, but their interaction with dietary iron absorption needs rigorous evaluation.
Tis review comprehensively synthesizes the burgeoning evidence linking ferroptosis, an iron-dependent form of regulated cell death, with the multifaceted process of aging. Our review highlights that ferroptosis is not merely a consequence of aging but an active contributor to age-related pathologies, intricately interwoven with established hallmarks such as oxidative stress, mitochondrial dysfunction, chronic infammation, and dysregulated iron metabolism. We have explored how age-associated declines in antioxidant defenses, particularly glutathione and GPx4, alongside paradoxical iron dynamics, create a fertile ground for ferroptosis activation.
Further delving into the mechanisms, the dysregulation of iron homeostasis during aging, characterized by systemic iron defciency alongside intracellular iron accumulation, signifcantly contributes to the Fenton reaction-driven production of ROS and subsequent lipid peroxidation. Tis interplay underscores ferroptosis as a critical
therapeutic target. Te natural products reviewed herein—such as resveratrol, curcumin, and EGCG—demonstrate remarkable pleiotropic mechanisms of action. Tey not only chelate excess iron and scavenge ROS but also upregulate endogenous antioxidant systems like Nrf2mediated pathways, thereby directly interfering with key ferroptotic drivers.
Te therapeutic potential of modulating ferroptosis in aging extends beyond single pathway inhibition. Our fndings suggest that strategies focusing on combinatorial regimens, perhaps pairing Nrf2 activation with iron chelation or lipid peroxidation inhibitors, could ofer synergistic benefts in mitigating age-related tissue damage. Future research should prioritize deciphering the precise molecular targets and signaling pathways of these natural compounds within specifc aging tissues and validating their efcacy through robust clinical trials with clear biomarkers of ferroptosis inhibition. Ultimately, the precise targeting of ferroptosis pathways through natural interventions holds signifcant promise as a novel strategy to promote healthy aging and prevent age-related diseases.
4. Conclusion
Ferroptosis ofers a unifying mechanism for oxidative stress, infammation, and metabolic dysfunction in aging. Natural products, with their ability to target multiple nodes of ferroptosis, present a compelling alternative to mitigate agerelated decline. While there are few studies that have shown the therapeutic potential of natural products in mitigating ferroptosis, and hence, age-related pathologies, integration of these compounds into therapeutic regimens could redefne aging interventions, shifting the paradigm from disease treatment to proactive antiaging efects.
4.1. Strengths and Limitations. Tis review provides a comprehensive and timely synthesis of the emerging understanding of ferroptosis in the context of aging and explores the therapeutic potential of natural products. Key strengths include:
- • A thorough literature survey integrating ferroptosis with established hallmarks of aging.
- • Detailed summarization of current animal studies and clinical trials related to natural products and ferroptosis in aging (as presented in tables).
- • Identifcation and elucidation of the mechanisms of action for various natural products in modulating ferroptosis.
- • Highlighting the potential for novel nutraceutical lead compounds or health supplements for the aging population.
Despite these strengths, certain limitations in the current understanding and research warrant consideration:
• Challenges persist in optimizing the bioavailability and delivery of many natural products, which can impact their efcacy in modulating ferroptosis in vivo.
- • Further research is needed to address tissue-specifc iron dysregulation associated with aging and the use of these natural products, as iron metabolism can vary signifcantly across diferent organs.
- • While preclinical data is promising, more robust and larger-scale human clinical trials are required to defnitively establish the therapeutic efcacy and safety of these natural compounds for ferroptosis inhibition in aging populations.
- • Te precise molecular targets and comprehensive signaling pathways by which all natural products exert their anti-ferroptotic efects are still being elucidated, necessitating further mechanistic studies.
Figures
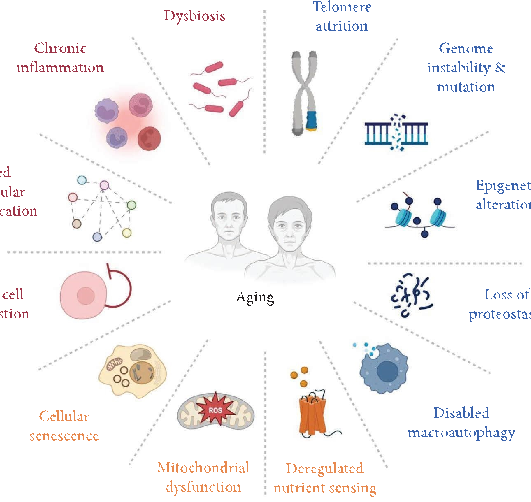
Figure 1
Conceptual diagram summarizing the relationship between modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products and the biological processes described in this research.
diagram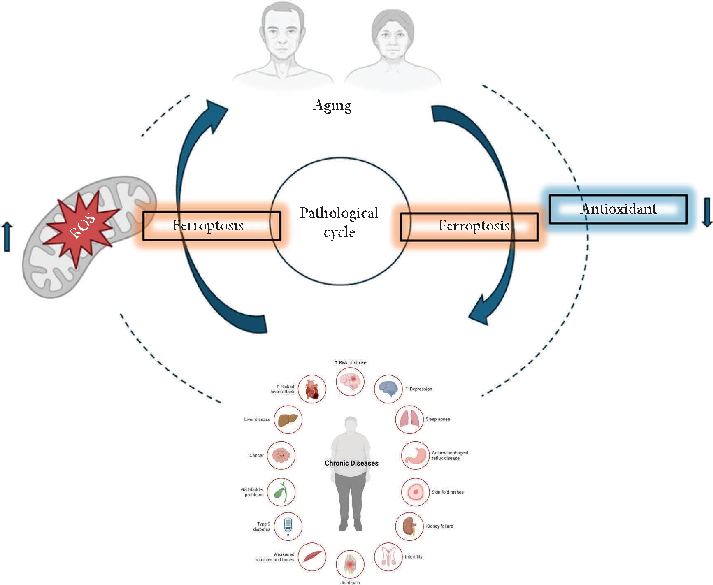
Figure 2
Visual summary of the pathways and interactions relevant to modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products, as discussed in the context of aging is a multifactorial process driven by accumulating cellular damage.
diagram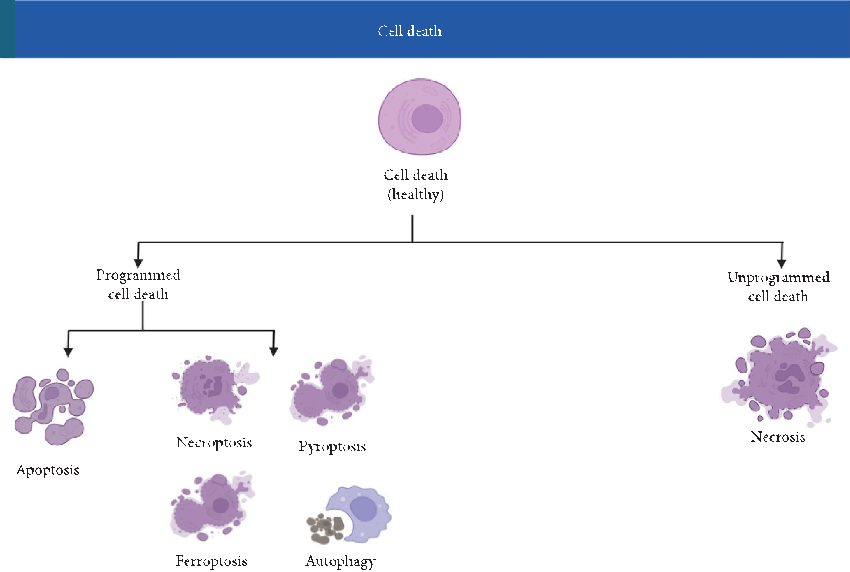
Figure 3
Schematic representation highlighting the mechanisms underlying modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products and their potential therapeutic implications.
diagram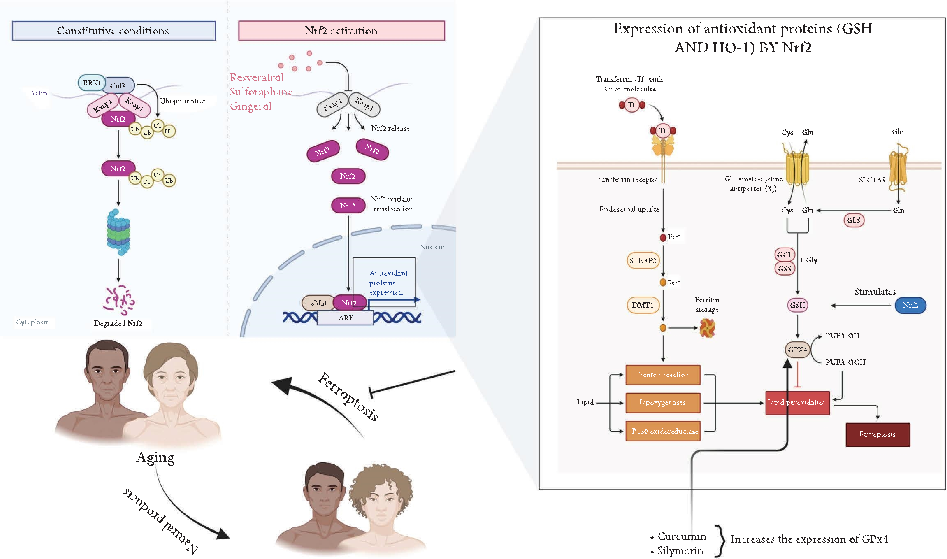
Figure 16
Experimental results examining modulating Ferroptosis in Aging: The Therapeutic Potential of Natural Products, with data points illustrating key findings related to aging is a multifactorial process driven by accumulating cellular damage.
chartUsed In Evidence Reviews
Similar Papers
Molecules (Basel, Switzerland) · 2015
Curcumin, inflammation, and chronic diseases: how are they linked?
BioFactors (Oxford, England) · 2013
Curcumin in inflammatory diseases.
Drug discovery today · 2017
Curcumin and its topical formulations for wound healing applications.
Seminars in cancer biology · 2022
Curcumin and colorectal cancer: An update and current perspective on this natural medicine.
International journal of molecular sciences · 2018
Anti-Inflammatory Effects of Resveratrol: Mechanistic Insights.
Biomedicines · 2021