Synergic Efficacy of a Multicomponent Nutraceutical Add-On Therapy in Seasonal Allergic Rhinitis in Children: A Prospective, Randomized, Parallel-Group Study.
Study Design
- Studientyp
- Randomized Controlled Trial
- Stichprobengröße
- 100
- Population
- Children with mild/moderate AR sensitized to grass pollens
- Dauer
- 12 weeks
- Intervention
- Synergic Efficacy of a Multicomponent Nutraceutical Add-On Therapy in Seasonal Allergic Rhinitis in Children: A Prospective, Randomized, Parallel-Group Study. Quertal + antihistamines
- Vergleichsgruppe
- Antihistamines alone
- Primärer Endpunkt
- Nasal/exhaled FeNO, nasal eosinophils
- Wirkungsrichtung
- Positive
- Verzerrungsrisiko
- Moderate
Abstract
Background: Emerging evidence suggests that nutraceuticals, alongside standard therapy, may benefit children with allergic rhinitis (AR). This study aimed to compare the efficacy of Quertal® (Neopharmed Gentili S.p.A., Milano, Italy), a nutraceutical supplement based on Perilla frutescens, Quercetin, and vitamin D3, combined antihistamines per os versus antihistamines alone, in improving AR symptoms considering respiratory functional and laboratory biomarkers in pediatric age. Materials and Method: This study included 100 children, 50 in the case group (Quertal® plus antihistamines) and 50 in the control group (antihistamines alone), with mild/moderate AR sensitized to grass pollens. They underwent assessments of respiratory function (rhinomanometry-AAR, spirometry), inflammation markers (Nasal Nitric Oxide [nFeNO]; exhaled Nitric Oxide [eFeNO]; nasal cytology), and laboratory assays (blood eosinophils, total IgE and specific IgE to Phl p1). Results: After three months of treatment, the case group showed statistically significant improvement in nFeNO and eFeNO values compared to controls (p < 0.001), as well as a reduction in nasal eosinophils (p < 0.001). Conclusions: Adding Quertal® to standard antihistamine therapy may reduce nasal inflammation and improve AR symptoms in pediatric patients. This combination therapy shows promise as a practical, well-tolerated approach to managing AR and may have broader implications for enhancing long-term outcomes.
Zusammenfassung
Adding Quertal® to standard antihistamine therapy may reduce nasal inflammation and improve AR symptoms in pediatric patients and may have broader implications for enhancing long-term outcomes.
Full Text
Synergic Efficacy of a Multicomponent Nutraceutical Add-On Therapy in Seasonal Allergic Rhinitis in Children: A Prospective, Randomized, Parallel-Group Study
Alessandra Gori *,† , Giulia Brindisi † , Caterina Anania , Alberto Spalice and Anna Maria Zicari
Department of Mother-Child, Urological Science, La Sapienza University, 00161 Rome, Italy; [email protected] (G.B.); [email protected] (C.A.); [email protected] (A.S.); [email protected] (A.M.Z.)
* Correspondence: [email protected] † These authors contributed equally to this work.
Academic Editor: Takao Fujisawa
Received: 23 January 2025 Revised: 11 February 2025 Accepted: 19 February 2025 Published: 24 February 2025
Citation: Gori, A.; Brindisi, G.; Anania, C.; Spalice, A.; Zicari, A.M. Synergic Efficacy of a Multicomponent Nutraceutical Add-On Therapy in Seasonal Allergic Rhinitis in Children: A Prospective, Randomized, Parallel-Group Study. J. Clin. Med. 2025, 14, 1517. https://doi.org/ 10.3390/jcm14051517
Copyright: © 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/ licenses/by/4.0/).
Abstract: Background: Emerging evidence suggests that nutraceuticals, alongside standard therapy, may benefit children with allergic rhinitis (AR). This study aimed to compare the efficacy of Quertal® (Neopharmed Gentili S.p.A., Milano, Italy), a nutraceutical supplement based on Perilla frutescens, Quercetin, and vitamin D3, combined antihistamines per os versus antihistamines alone, in improving AR symptoms considering respiratory functional and laboratory biomarkers in pediatric age. Materials and Method: This study included 100 children, 50 in the case group (Quertal® plus antihistamines) and 50 in the control group (antihistamines alone), with mild/moderate AR sensitized to grass pollens. They underwent assessments of respiratory function (rhinomanometry-AAR, spirometry), inflammation markers (Nasal Nitric Oxide [nFeNO]; exhaled Nitric Oxide [eFeNO]; nasal cytology), and laboratory assays (blood eosinophils, total IgE and specific IgE to Phl p1). Results: After three months of treatment, the case group showed statistically significant improvement in nFeNO and eFeNO values compared to controls (p < 0.001), as well as a reduction in nasal eosinophils (p < 0.001). Conclusions: Adding Quertal® to standard antihistamine therapy may reduce nasal inflammation and improve AR symptoms in pediatric patients. This combination therapy shows promise as a practical, well-tolerated approach to managing AR and may have broader implications for enhancing long-term outcomes.
Keywords: allergic rhinitis (AR); nutraceuticals; Quercetin; Perilla frutescens; vitamin D3; nasal nitric oxide; exhaled nitric oxide; nasal cytology; randomized controlled trial (RCT); pediatric allergy
1. Introduction
Allergic rhinitis (AR) is a prevalent chronic respiratory disease affecting up to 50% of the global population, particularly in industrialized countries. It poses a significant public health concern due to its social and economic impact [1]. In children, AR manifests with high frequency (from 10% to 30% incidence [2], while prevalence rises as children grow older, from 8.5% to 14.6% (ISAAC) [3,4]).
The delayed diagnosis or improper management can lead to the persistence and progression of symptoms from the upper to the lower respiratory tract, ultimately causing chronic conditions [3,5,6].
Despite advancements in understanding AR’s pathogenesis, based on classic Type
2 inflammation that involved a complex architecture of cellular and humoral mediators
J. Clin. Med. 2025, 14, 1517 https://doi.org/10.3390/jcm14051517
enabling the cascading recruitment and bridging of innate and adaptive immunity [7–9], there remains a pressing need for innovation in treatment options. Current management largely depends on symptomatic therapy, such as antihistamines and corticosteroids, and Allergen-specific Immunotherapy (AIT), the only disease-modifying management option [10]. International researchers’ interest in the bioactive principles of complementary medicine has led to the utilization of nutraceuticals, offering new therapeutic possibilities. This approach is based on applying current knowledge on the pathogenesis of diseases and verifying the efficacy of such compounds through scientific methods and a careful process of validation, transformation, and production [11].
Among the most notable nutraceuticals mentioned in the literature in this field, Quertal® leverages bioactive compounds from food and medicinal plants to target the inflammatory pathways of AR, potentially enhancing the efficacy of standard treatments while reducing their side effects. Containing Perilla frutescens, Quercetin, and vitamin D3, this supplement stands out for its anti-inflammatory, antioxidant, and immunomodulatory properties due to its unique composition [12–14]. In particular, in Perilla frutescens, 271 active compounds have been identified in the seeds, stems, and leaves, including rosmarinic acid and luteolin with anti-allergic (antihistaminic with inhibition of mast cell degranulation); antioxidant (scavenging of ROSs (suppression of inducible nitric oxide synthase (iNOS) expression)); and anti-inflammatory (reduction in the release of the HMGB1 protein with an inhibitory effect on pro-inflammatory cytokines (TNF-α and IL-6) and Nitric Oxide) properties [12,15,16].
Quercetin is the most abundant dietary flavonoid, present in a wide range of fruits, vegetables, and beverages, with high interest and success in the literature (currently over 23,000 citations in PubMed). From this vast number of data, quercetin improves pathological conditions involving inflammation and oxidative stress at different levels of the allergic inflammatory cascade by acting on cellular and humoral targets throughout the respiratory tree. In particular, Quercetin reduces airway allergic inflammation and hyperreactivity caused by the Th1/Th2 imbalance and has a suppressive effect on the cells’ ability to produce CCL5, eotaxin, periostin, and NO, leading to an improvement in the clinical condition of patients with AR [14,17–21].
Indeed, the last compound is vitamin D3. Its role suggests a dynamic process that begins in the womb and continues throughout childhood and probably into adulthood, during which vitamin D plays a critical role in interfering with the immunopathogenic mechanisms of respiratory allergy development. The increase in allergic diseases has coincided with the frequent deficiency represented in many populations of vitamin D, and this inverse relationship has raised the idea of a possible underlying association between these two opposite trends [13].
Vitamin D impacts both innate and adaptive immunity by inhibiting Toll-like receptors on monocytes, reducing proinflammatory cytokines, and promoting antimicrobial peptides. It influences T and B lymphocytes, reducing IgE secretion, balancing Th1/Th2 lymphocytes, and supporting regulatory T cells (Treg). Vitamin D deficiency can shift this balance, leading to increased Th2 response. It also affects airway remodeling and maintains nasal epithelium integrity, protecting against allergens. Studies show that vitamin D enhances the response to dexamethasone by improving IL-10 production and reducing steroid resistance in asthma [22].
Thus, this study aims to evaluate the potential of non-pharmacological treatments to complement conventional therapies in managing AR in children. By using tools like anterior active rhinomanometry (AAR), spirometry, nasal and exhaled nitric oxide (nFeNO, eFeNO), nasal cytology, and laboratory tests (eosinophils, total and specific IgE levels), it
provides evidence for improved prevention, particularly in secondary and tertiary care, offering tailored therapeutic strategies for pediatric patients.
2. Materials and Methods
- 2.1. Study Design
- 2.2. Study Population
- 2.3. Randomization
- 2.4. Study Phases
- 1. T0-Enrollment phase: This phase involved allergological visit and demographic
and anamnestic data following inclusion/exclusion criteria. Then, a physical examination, a clinical evaluation with the classification of AR according to ARIA criteria, and Skin Prick Tests (SPTs) were performed. Suitable patients were then allocated in a 1:1 ratio into two groups: The case group treated with Quertal® double-layer tablets (1 tablet per day
for three months) plus oral antihistamines therapy, and the control group treated with oral antihistamines only.
- 2. T1-Start of therapy: One week approximately after T0, a clinical evaluation was
conducted at a day hospital (DH), where laboratory and instrumental tests were performed. Therapy was prescribed (Quertal® plus antihistamines for the case group, antihistamines only for the control group), and patients were given clinical diaries to record symptoms, occurrence/duration of exacerbations, and adverse events.
3. T2-Follow-up: After three months of therapy administration, another clinical evaluation was conducted at the DH, repeating the laboratory and instrumental tests performed at T1.
- 2.5. Study Outcomes
- 2.6. Serological Biomarkers
- 2.7. Nasal Eosinophils Count and C/G Ratio at Nasal Cytology
- 2.8. Nasal Nitric Oxide (nFeNO)
Nasal FeNO (nFeNO) was performed in spontaneous breathing and using a tight facemask with a fixed flow connected to an analyzer (Medisoft nFeNO analyzer, Medisoft s.r.l., Naples, Italy). The measurement of nFeNO was made according to the American Thoracic Society/European Respiratory Society guidelines’ values (ERS/ATS). During inspiration up to total lung capacity, patients inhaled through the nose from the NO-free analyzer and exhaled through disposable nose pads at a constant flow of 350 mL/s for 60 s [25,26].
- 2.9. Exhaled Nitric Oxide (eFeNO)
- 2.10. Mean Nasal Flow (mNF) at Active Anterior Rhinomanometry (AAR)
- 2.11. Forced Expiratory Volume in the First Second (FEV1) at Spirometry
- 2.12. Nasal Symptom Score (NSS)
- 2.13. Treatment Compliance, Rescue Medication, and Adverse Events
- 2.14. Schedule of Quertal® Administration
Regarding the method of administration, each patient of the “case group” took one tablet of Quertal® a day, administered together with antihistamine therapy for three months.
Each tablet had a dual layer for fast Perilla and slow Quercetin/vitamin D3 release. The specific content of the active ingredients is 80 mg for Perilla frutescens, 150 mg for Quercetin, and 5 mcg (200 IU) for vitamin D3.
- 2.15. Standard Treatment
Standard therapy was an antihistamine chosen by the investigator, considering the specific needs of the patients. Generally, Cetirizine or Levocetirizine were the preferable antihistamine molecules.
3. Statistical Analysis
The sample size was determined based on a power calculation to detect a clinically significant difference in nasal nitric oxide levels (nFeNO) between groups. A hundred patients (50 per group) were required to achieve an 80% power with an alpha level of 0.05. This calculation was based on previous studies assessing similar nutraceutical interventions in allergic rhinitis. Statistical analysis was performed using IBM SPSS version 27.0 (SPSS, Chicago, IL, USA). For the continuous variables, the Shapiro–Wilk normality test was performed; these continuous quantities were represented by the mean value and standard deviation (SD). Nominal and ordinal variables were described in terms of relative counts and frequencies. The comparison of the assumed clinical values—both for the case and control groups—was performed by “paired-samples T-test” and by “Wilcoxon signed-rank test for paired samples”. The comparison between the case and control groups, both at T0 and at T1, was performed by the “t-test for unpaired samples” and the “U Mann–Whitney test”. In all cases, a p-value ≤ 0.05 was considered statistically significant.
4. Results
The characteristics of the study population are reported in Table 1 below. No significant differences were noted regarding age, weight, height, and BMI between the case and control groups. The associated presence of mild intermittent asthma between the case and control groups is reported in Table 1.
Table 1. Characteristics of the study population. Allergic Rhinitis (AR).
Characteristics Mean ± SD p-Value
Age (years)
- - Control 9.52 ± 2.81 p = 0.653
- - Case 8.99 ± 3.08
Weight (kg)
- - Control 35.67 ± 13.94 p = 0.573
- - Case 34.15 ± 13.01
Height (cm)
- - Control 135.76 ± 16.75 p = 0.423
- - Case 133.06 ± 16.80
BMI (kg/m2)
- - Control 18.59 ± 3.33 p = 0.938
- - Case 18.64 ± 3.63
Characteristics Count Percent Sex (Female)
- - Control 18 36%
- - Case 20 40% AR
- - Control 50 100%
- - Case 50 100%
- Table 1. Cont. Characteristics Mean ± SD p-Value
- Table 2. Comparison of intragroup and intergroup values related to the considered variables between the case and control. Mean Nasal Flow (mNF); Nasal Nitric Oxide (nFeNO); Forced Expiratory Volume in the First Second (FEV1); Exhaled nitric oxide (eFeNO); and ratio of ciliated cells to goblet cells (C/G Ratio).
Value
p-Value Intragroup
T0 T1 Mean ± SD
Characteristic
p-Value Intergroup Nasal Symptom Score (NSS)
p-Value Intergroup
Mean ± SD
- - Control 22.10 ± 4.76 p = 0.911
- - Case 22.00 ± 4.12 13.38 ± 3.61 p < 0.001
mNF (%) pre-hydrazine
- - Control 67.42 ± 25.98 p = 0.957
- - Case 67.14 ± 26.18 80.60 ± 20.34 p = 0.001
mNF (%) post-hydrazine
- - Control 74.25 ± 22.53 p = 0.872
- - Case 74.95 ± 20.63 86.72 ± 15.81 p = 0.001
Nasal FeNO (nFeN0)
- - Control 1275 ± 660.69 p = 0.985
- - Case 1277.5 ± 697.21 798.10 ± 436.21 p = 0.001
- - Control 99.70 ± 9.38 p = 0.763
- - Case 100.30 ± 10.46 101.02 ± 11.37 p = 0.104
- - Control 103.24 ± 9.27 p = 0.920
- - Case 103.44 ± 10.54 105.48 ± 12.34 p = 0.067
Exhaled FeNO
- - Control 14.88 ± 5.43 p = 0.987
- - Case 14.86 ± 3.79 10.33 ± 3.36 p < 0.001
Blood Eosinophils
- - Control 5.65 ± 3.61 p = 0.993
- - Case 5.64 ± 2.73 5.34 ± 2.50 p = 0.121
Nasal Eosinophils
- - Control 9.00 ± 3.64 p = 0.739
- - Case 9.20 ± 2.16 6.20 ± 2.06 p = 0.001
‐ Control 14.88 ± 5.43 14.89 ± 4.46 p = 0.979
- J. Clin. Med. 2025, 14, 1517 8 of 18
‐ Control
p = ‐ Case 5.64 ± 2.73 5.34 ± 2.50 p =
p = 0.993 p = 0.625
Table 2. Cont.
p = 0.739 p = 0.001 Total IgE,Ku/L
Value
p-Value Intragroup
T0 T1 Mean ± SD
Characteristic
322.18 ± 271.77
322.27 ± 271.81
p-Value Intergroup Total IgE, Ku/L
p-Value Intergroup
Mean SD
p =
p = 0.992
‐ Case 322.73 ± 192.74 308.88 ± 172.79 p = C/G Ratio
- - Control 322.27 ± 271.81 p = 0.992
- - Case 322.73 ± 192.74 308.88 ± 172.79 p = 0.058
‐ Control 1.30 ± 0.60
1.29 ± 0.55
0.938
0.001
C/G Ratio
‐ Case Phl p1
- - Control 1.30 ± 0.60 p = 0.938
- - Case 1.31 ± 0.68 2.72 ± 1.34 p = 0.001 Phl p1
- - Control 14.77 ± 9.75 p = 0.583
- - Case 15.89 ±10.53 14.70 ± 9.03 p = 0.093
‐ Control 14.77 ± 9.75 14.63 ± 9.15
0.968
‐ Case 15.89 ±10.53 14.70 ± 9.03
Considering the NSS intragroup variability both for the case and control groups, found a statistically significant difference between T0 and T1 values (p < 0.001). The same significance for the NSS intergroup variability at T1 (p < 0.001) was found, as shown in box plot Figure 1 and Table 2.
Considering the NSS intragroup variability both for the case and control groups, we found a statistically significant difference between T0 and T1 values (p < 0.001). The same significance for the NSS intergroup variability at T1 (p < 0.001) was found, as shown in the box plot Figure 1 and Table 2.
Figure 1. Nasal symptom score (NSS) at T0 and T1 in the case and control groups. Box plot representing NSS values at T0 and T1 in both the case and control groups (** significant).
Figure 1. Nasal symptom score (NSS) at T0 and T1 in the case and control groups. Box plot repre senting NSS values at T0 and T1 in both the case and control groups (** significant).
Considering mNF pre-hydrazine and mNF post-hydrazine, we found statistical significance for the intragroup variability both for the case and control groups, between T0 and T1 values (p < 0.001). The same significance was found for the intergroup variability at T1, as shown in Figure 2a,b and Table 2.
Considering mNF pre‐hydrazine and mNF post‐hydrazine, we found statistical sig nificance for the intragroup variability both for the case and control groups, between and T1 values (p < 0.001). The same significance was found for the intergroup variability at T1, as shown in Figure 2a,b and Table 2.
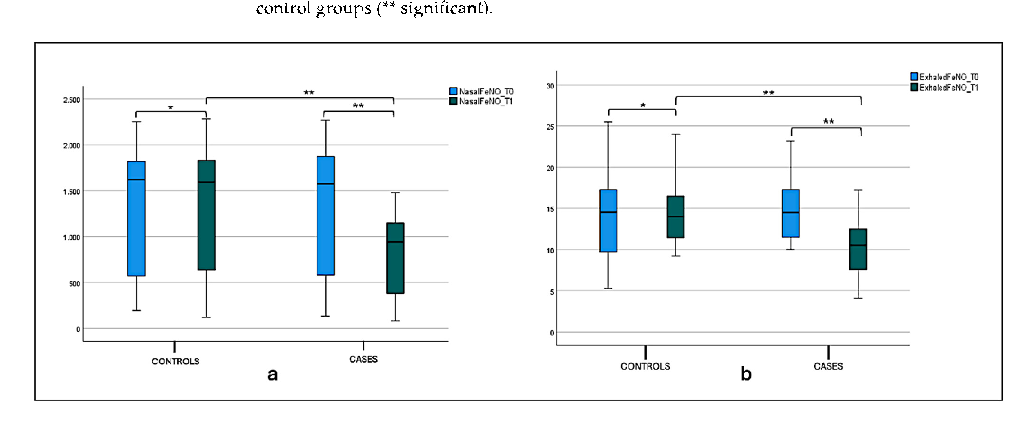
A statistical significance difference between nFeN0 at T0 (1277.5 ± 697.21) and T1 (798.1 ± 436.21) (p < 0.001) was observed in the case group, as shown in Figure 3, and Table 2 Considering the intergroup variability, we did not find a statistically significant difference between the nFeNO of the case and control groups at T0; this difference was found at T1 (p < 0.001), as shown in Figure 3a and Table 2. The eFeNO values in the case group showed a statistical significance difference between eFeN0 at T0 (14.86 ± 3.79) and T1 (10.33 ± 3.36) (p < 0.001), as shown in Figure 3b and Table 1. Considering the intergroup variability, we did not find a statistically significant difference between the eFeNO of the
- J. Clin. Med. 2025, 14, 1517 9 of 18
case and control groups at T0; this difference was found at T1 (p < 0.001), as shown in Figure 3b and Table 2.
, x FOR PEER REVIEW 9 of 19
(a) (b)
) Mean nasal flow (mNF) pre‐hydrazine . Box fl control
n control groups. Box plot represe both ificant).
e
< 0.001) was able 2 Considering the intergroup ffer‐ ence between the nFeNO of t und
< 0.001), as shown in oup showed a statistical significance difference between eFeN0 at T0 (14.86 ± 3.79) and
(a) (b)
- Figure 2. (a) Mean nasal flow (mNF) pre‐hydrazine at T0 and T1 in the case and control groups. Box plot representing mean nasal flow (mNF) pre‐hydrazine at T0 and T1 in both case and control groups (** significant). (b) Mean nasal flow (mNF) post‐hydrazine at T0 and T1 in the case and control groups. Box plot representing mean nasal flow (mNF) post‐hydrazine at T0 and T1 in both case and control groups (** significant).
- Figure 3. (a) Nasal FeNO (nFeNO) at T0 and T1 in the treatment and control groups. Box plot rep‐ resenting nFeNO values at T0 and T1 in both the treatment and control groups (* not significant, ** significant). (b) Exhaled FeNO (eFeNO) at T0 and T1 in the treatment and control groups. Box plot representing eFeNO values at T0 and T1 in both the treatment and control groups (* not significant,
Figure 2. (a) Mean nasal flow (mNF) pre-hydrazine at T0 and T1 in the case and control groups. Box plot representing mean nasal flow (mNF) pre-hydrazine at T0 and T1 in both case and control groups (** significant). (b) Mean nasal flow (mNF) post-hydrazine at T0 and T1 in the case and control groups. Box plot representing mean nasal flow (mNF) post-hydrazine at T0 and T1 in both case and control groups (** significant).
variability, we did not find a statistically significant difference between the eFeNO of the case and control groups at T0; this difference was found at T1 (p < 0.001), as shown in Figure 3b and Table 2.
Figure 3. (a) Nasal FeNO (nFeNO) at T0 and T1 in the treatment and control groups. Box plot representing nFeNO values at T0 and T1 in both the treatment and control groups (* not significant, ** significant). (b) Exhaled FeNO (eFeNO) at T0 and T1 in the treatment and control groups. Box plot representing eFeNO values at T0 and T1 in both the treatment and control groups (* not significant, ** significant).
Figure 3. (a) Nasal FeNO (nFeNO) at T0 and T1 in the treatment and control groups. Box plot rep‐ resenting nFeNO values at T0 and T1 in both the treatment and control groups (* not significant, ** significant). (b) Exhaled FeNO (eFeNO) at T0 and T1 in the treatment and control groups. Box plot representing eFeNO values at T0 and T1 in both the treatment and control groups (* not significant,
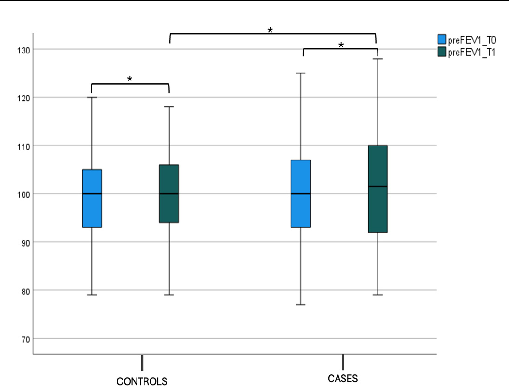
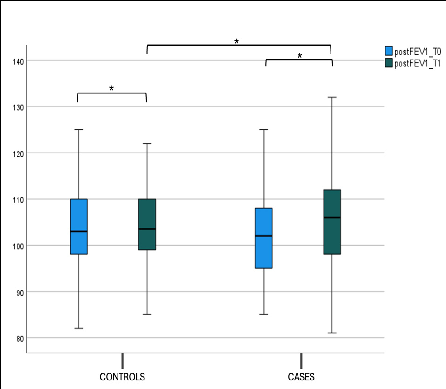
Instead, we did not find a statistically significant difference in both preFEV1 and postFEV1 values between the case and control groups at T1 (p-value intergroup). This difference was not found, respectively, for “preFEV1” and “postFEV1” intragroup values, as shown in Figure 4a,b and Table 2.
Considering the percentage of eosinophil cells in nasal cytology, we found a statistical significance at T0 (9.20 ± 2.16) and T1 (6.20 ± 2.06) (p < 0.001) in the case group, as shown in Figure 5a and Table 2. We did not find a statistically significant difference in the percentage of eosinophil cells between the case and control groups at T0; this difference was found at T1 (p < 0.001), as shown in Figure 5a and Table 2. The Cilateted/Goblet cells (C/G ratio) in the case group showed a statistical significance at T0 (1.31 ± 0.68) and T1 (2.72 ± 1.34)
- J. Clin. Med. 2025, 14, 1517 10 of 18
(p < 0.001), as shown in Figure 5b and Table 1. We did not find a statistically significant difference in the C/G ratio between the case and control groups at T0, while this difference was found at T1 (p < 0.001) (Figure 5b and Table 2).
postFEV1 values between the case and control groups at T1 (p‐value intergroup). This difference was not found, respectively, for “preFEV1” and “postFEV1” intragroup values, as shown in Figure 4a,b and Table 2.
- (a) (b)
- (b) FEV1 post‐bronchodilatation (postFEV1) at T0 and T1 in the case and control groups. Box plot representing postFEV1 values at T0 and T1 in both the case and control groups (* not significant).
Considering the percentage of eosinophil cells in nasal cytology, we found a statisti‐ cal significance at T0 (9.20 ± 2.16) and T1 (6.20 ± 2.06) (p < 0.001) in the case group, as shown in Figure 5a and Table 2. We did not find a statistically significant difference in the per‐ centage of eosinophil cells between the case and control groups at T0; this difference was found at T1 ( < 0.001), as shown in Figure 5a and Table 2. The Cilateted/Goblet cells (C/G ratio) in the case group showed a statistical significance at T0 (1.31 ± 0.68) and T1 (2.72 ± 1.34) (p < 0.001), as shown in Figure 5b and Table 1. We did not find a statistically signifi‐ cant difference in the C/G ratio between the case and control groups at T0, while this dif‐ ference was found at T1 (p < 0.001) (Figure 5b and Table 2).
- (a) (b)
- (b) FEV1 post‐bronchodilatation (postFEV1) at T0 and T1 in the case and control groups. Box plot representing postFEV1 values at T0 and T1 in both the case and control groups (* not significant).
Figure 4. (a) FEV1 pre-bronchodilatation (preFEV1) at T0 and T1 in the case and control groups. Box plot representing preFEV1 values at T0 and T1 in both the case and control groups (* not significant). (b) FEV1 post-bronchodilatation (postFEV1) at T0 and T1 in the case and control groups. Box plot representing postFEV1 values at T0 and T1 in both the case and control groups (* not significant).
Considering the percentage of eosinophil cells in nasal cytology, we found a statisti‐ cal significance at T0 (9.20 ± 2.16) and T1 (6.20 ± 2.06) ( < 0.001) in the case group, as shown in Figure 5a and Table 2. We did not find a statistically significant difference in the per‐ centage of eosinophil cells between the case and control groups at T0; this difference was found at T1 ( < 0.001), as shown in Figure 5a and Table 2. The Cilateted/Goblet cells (C/G ratio) in the case group showed a statistical significance at T0 (1.31 ± 0.68) and T1 (2.72 ± 1.34) (p < 0.001), as shown in Figure 5b and Table 1. We did not find a statistically signifi‐ cant difference in the C/G ratio between the case and control groups at T0, while this dif‐ ference was found at T1 (p < 0.001) (Figure 5b and Table 2).
- (a) (b)
- (b) C/G ratio at T0 and T1 in the case and control group. Box plot representing C/G ratio at T0 and T1 in both case and control group (* not significant, ** significant).
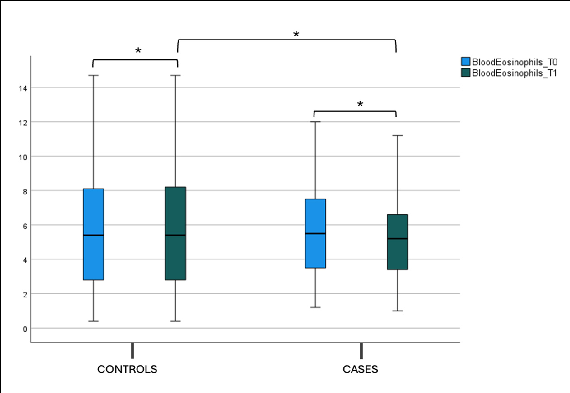
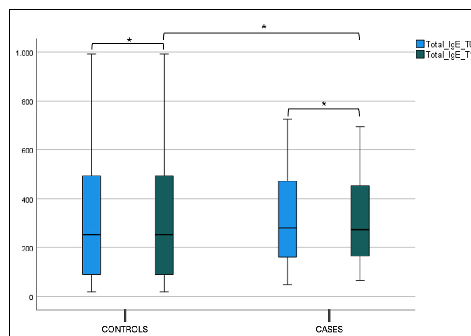
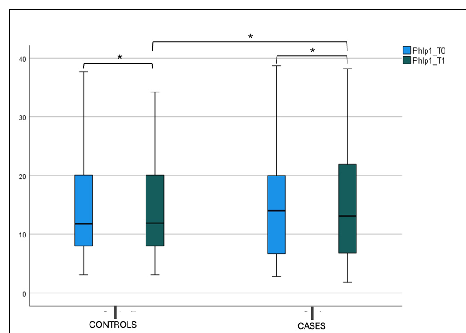
No statistical difference was found in the blood eosinophils count, total IgE (tIgE), and Phlp1 values in the control and case groups at T0 and T1 in both the intergroup and intragroup variability (Figure 6, Figure 7a,b and Table 2).
J. Clin. Med. 2025, 14, 1517 Phlp1 values in the control and case groups at T0 and T1 in both the intergroup and in‐11 of 18
- Figure 6. Blood eosinophils count at T0 and T1 in the case and control groups. Box plot representing eosinophils values at T0 and T1 in both the case and control groups (* not significant).
- Figure 7. (a) Total IgE (tIgE) level at T0 and T1 in the case and control group. Box plot representing total IgE (tIgE) level at T0 and T1 in both case and control group (* not significant). (b) Phlp1 level at T0 and T1 in the case and control group. Box plot representing Phlp1 level at T0 and T1 in both case and control group (* not significant).
Figure 6. Blood eosinophils count at T0 and T1 in the case and control groups. Box plot representing eosinophils values at T0 and T1 in both the case and control groups (* not significant).
Figure 7. (a) Total IgE (tIgE) level at T0 and T1 in the case and control group. Box plot representing total IgE (tIgE) level at T0 and T1 in both case and control group (* not significant). (b) Phlp1 level at T0 and T1 in the case and control group. Box plot representing Phlp1 level at T0 and T1 in both case and control group (* not significant).
5.
5. Discussion
The encouraging findings of this study demonstrate that the addition of Quertal to antihistamine therapy significantly improved both subjective and objective markers of mild/moderate AR in children. Specifically, the combination of Quertal with
The encouraging findings of this study demonstrate that the addition of Quertal® to antihistamine therapy significantly improved both subjective and objective markers of mild/moderate AR in children. Specifically, the combination of Quertal® with antihistamines resulted in significant reductions in both nasal and exhaled nitric oxide levels (nFeNO and eFeNO), which are widely recognized markers of allergic inflammation. Additionally, the treatment group also saw a marked decrease in nasal eosinophil counts, further validating Quertal’s role as an anti-inflammatory agent.
After three months of treatment, patients in the Quertal® group exhibited a 30% reduction in nFeNO levels from baseline, compared to a 5% decrease in the control group. Likewise, eFeNO levels dropped by 25% in the Quertal® group, significantly reducing allergic inflammation. Nasal eosinophil counts were also notably reduced by 40%, suggesting that the nutraceutical may be influential in managing eosinophil-mediated inflammation. Nasal eosinophil counts were also reduced by 40%, suggesting that the nutraceutical may
influence eosinophil-mediated inflammation. The significant improvement in the C/G ratio confirms the amelioration of nasal cellularity [31,32]. Goblet cell hyperplasia is a hallmark of AR, characterized by an increased number of goblet cells in the nasal mucosa. This increase is associated with the inflammatory response and tissue remodeling processes, including eosinophil infiltration and epithelial damage [33]. In addition, post-therapy, the mean nasal flow improved by 20%.
The choice of cetirizine or levocetirizine as the antihistamine in this study plays a practical role in the observed outcomes. These second-generation antihistamines, in addition to being widely available, are well known for reducing symptoms such as nasal congestion, sneezing, and itching, exacerbated by grass pollen allergens [34–37]. The synergistic effect between antihistamines and Quertal®, mainly in reducing nasal eosinophil recruitment and cytokine release, likely accounts for the significant improvements in both nFeNO and eFeNO levels in the case group.
The specific formulation of Quertal® may significantly contribute to this synergistic effect, particularly considering that the lipid matrix based on medium-chain triglycerides in the slow-release layer improves the bioavailability of Quercetin. However, the inherent challenges of Quercetin’s rapid metabolism, low solubility, and efflux mechanisms that limit its overall bioavailability still need a thorough examination. In the case of Perilla frutescens, it may contribute to its anti-inflammatory and anti-allergic effects, but proper standardization would ensure that each supplement dose contains an adequate and predictable amount of its compounds, and their bioavailability would be adequately addressed.
In this context, the decision to exclude intranasal corticosteroids played a crucial role in isolating the specific effects of Quertal® because their potent anti-inflammatory effect could alter the results of nasal cytology. Additionally, by avoiding intranasal corticosteroids that significantly reduce nitric oxide (NO) levels, the study was able to demonstrate that the supplement itself may substantially reduce inflammatory markers like nFeNO and eFeNO.
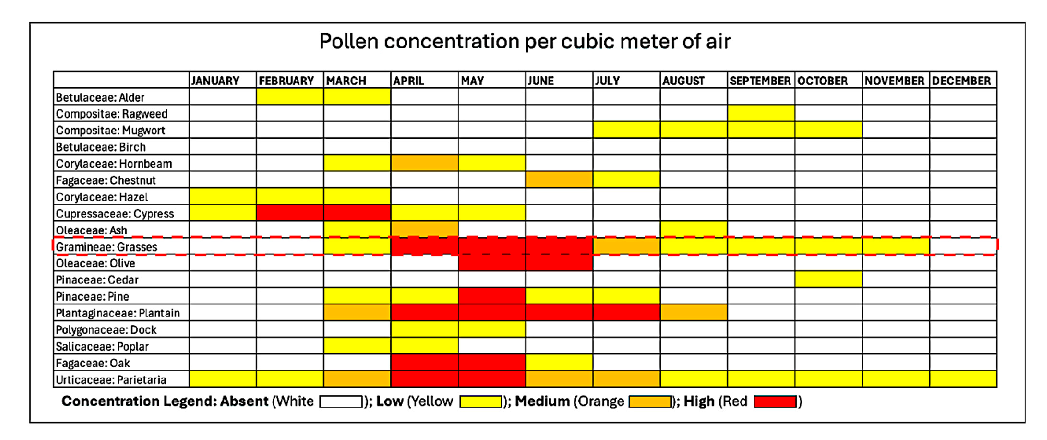
Monosensitization to Phleum pratense (Timothy grass), one of the most prevalent allergens in urban areas like Rome, was considered to further target the nutraceutical’s effect. The well-defined pollination season of grass pollen, peaking in spring to early summer, allowed us to focus the study on a specific period when AR symptoms are at their onset until most pronounced, as demonstrated in Figure 8. In our opinion, this approach ensured a consistent timeframe for evaluating a preventive effect in reducing symptoms and treatment efficacy, while avoiding confounding factors such as new sensitization to overlapping pollination.
Our results align with previous studies by including pediatric patients and focusing on subjective (NSS) and objective (AAR, nasal cytology) markers, providing a more comprehensive view of Quertal®’s benefits. Unlike previous studies that primarily relied on subjective symptom relief, our work emphasizes quantifiable inflammatory markers, offering a more robust evaluation of the nutraceutical’s effects.
Many studies attest to the effectiveness of the association of these molecules in the management of the clinical symptoms of AR and in reducing the need for drugs in the pediatric age.
Through such an Italian clinical trial, it was demonstrated in the first phase that the multicomponent nutraceutical (Lertal®) improved the effect of antihistamine treatment with an average reduction of about 64% in AR symptoms’ severity, thus possibly reducing the onset of clinical relapses with savings on drug use. The second phase of the same trial, a parallel-group extension study lasting 4–12 weeks, in which cases continued treatment with the nutraceutical tablets, confirmed the favorable effects of the first phase, halving the risk of AR exacerbation after one month of antihistamine treatment and with a preventive effect towards clinical exacerbations [38,39]. There were also demonstrated changes over time in
spirometry after a year, a result that could mean a potential prophylactic impact towards the possible onset of asthma in patients with AR. In our study, spirometric data (FEV1) at baseline and post-bronchodilation did not present statistically significant differences. However, the enrolled patients had no clinically evident asthma symptoms, and the spirometric values were average, so we did not find any changes in the pre- and post-therapy spirometric values. However, in general, better control of AR can significantly improve the spirometric picture, as well as the bronchial inflammation, as seen in our group of patients by the significant reduction in eFeNO values. So, a reasonable control of the AR improves the eFeNO values, even in patients who did not have positive values of the spirometric parameters and, therefore, who present a chronic basal bronchial inflammation not yet clinically manifest with positive spirometry [25,40].
, x FOR PEER REVIEW 13 of 19
Figure 8. Pollen calendar of the metropolitan area of Rome. The red dotted line encloses Grass Pollen concentration, which our patients were exclusively sensitized to, in the metropolitan area of Rome from which all our patients resided. The pollen calendar was developed by processing a multi-year data series collected by the Aerobiological Monitoring Center of the University of Rome Tor Vergata. For each family, three concentration levels are indicated: low, medium, and high, represented by yellow, orange, and red, respectively. The calendar is available at https://polline. uniroma2.it/calendario-pollinico-della-citta/. Accessed on 15 December 2022.
Figure 8. Pollen calendar of the metropolitan area of Rome. The red dotted line encloses Grass Pollen concentration, which our patients were exclusively sensitized to, in the metropolitan area of Rome from which all our patients resided. The pollen calendar was developed by processing a multi-year data series collected by the Aerobiological Monitoring Center of the University of Rome Tor Vergata. For each family, three concentration levels are indicated: low, medium, and high, represented by yellow, orange, and red, respectively. The calendar is available at https://polline.uni-
Another study recently evaluated the use of the same nutraceutical component as adjunctive therapy in children affected by AR, showing it to help reduce antihistamine use in the case group in comparison with the control group under standard treatment only [41].
Our results align with previous studies by including pediatric patients and focusing on subjective (NSS) and objective (AAR, nasal cytology) markers, providing a more comprehensive view of Quertal®’s benefits. Unlike previous studies that primarily relied on subjective symptom relief, our work emphasizes quantifiable inflammatory markers, offering a more robust evaluation of the nutraceutical’s effects.
The study conducted by Ariano tested 23 adults with positive SPTs to Parietaria to evaluate the efficacy of the Lertal® on symptoms of seasonal allergic rhinitis and the consumption of anti-allergic drugs. The improvement of nasal symptoms was tested using the Total Symptoms Score at the first (baseline) and second (final) visit. The conclusion showed an apparent efficacy of Lertal® in reducing nasal symptoms (70% symptoms, 73% use of antiallergic drugs, p < 0.001). This activity was objectively confirmed by reducing the consumption of anti-allergic medications to relieve symptoms. No side effects were reported. The critical lack of such a study was the control group, and the authors do not explain in detail what type of anti-allergic therapies are carried out [42]. Similar results of a reasonable control of AR symptoms were found in our study.
Many studies attest to the effectiveness of the association of these molecules in the management of the clinical symptoms of AR and in reducing the need for drugs in the pediatric age.
Through such an Italian clinical trial, it was demonstrated in the first phase that the multicomponent nutraceutical (Lertal®) improved the effect of antihistamine treatment with an average reduction of about 64% in AR symptoms’ severity, thus possibly reducing the onset of clinical relapses with savings on drug use. The second phase of the same trial, a parallel-group extension study lasting 4–12 weeks, in which cases continued treatment with the nutraceutical tablets, confirmed the favorable effects of the first phase, halving the risk of AR exacerbation after one month of antihistamine treatment and with a pre-
A recent study evaluated the possible change in spirometry in two groups of children affected by AR: The first group under a nutraceutical (Lertal®), and the second one was the control group. The children were visited at baseline and the end of the nutraceutical
treatment after one year. The results confirmed that children in the case group had a significantly higher MEF50 than the control group. In conclusion, the study demonstrated that a course with a multicomponent nutraceutical could prevent the decline of MEF50 in children with AR [43].
Instead, no changes were detected in blood parameters, such as blood eosinophil count, tIgE, and sIgE for Phlp1. A more prolonged study duration could have provided significant variations within the laboratory parameters.
Regarding blood parameters, a recent study conducted in a mouse model with AR using the ionic cross-linking method for intranasal administration of quercetin (QCS) demonstrated that QCS treatment significantly reduced the number of sneezes and nasal rubs in mice with AR while reducing the levels of inflammatory factors such as immunoglobulin E (IgE), interleukin (IL)-17, tumor necrosis factor (TNF)-α, and (IL)-6 to relieve the AR symptoms. Furthermore, thanks to hematoxylin-eosin (HE) staining, improvement of a damaged nasal mucosa has been demonstrated. These experimental results suggest that QCS can effectively suppress allergic inflammation in a mouse model and represent a promising therapeutic option for AR in humans [44]. Another recent study on a murine model was published on the anti-allergic biological activity of Quercetin in AR, where the authors aimed to explore the effects of Quercetin on the balance of T helper type 1 (Th1)/Th2 cells and regulatory T cells (Treg)/Th17. The results showed that Quercetin alleviates nasal itching and sneezing from a clinical point of view. It was also assessed through a laboratory evaluation that Quercetin reduced IgE and IgG1 and increased IgG2 in serum, inactivating the NF-κB pathway. Taken together, Quercetin attenuated AR symptoms by balancing the Th1/Th2 and Treg/Th17 ratios and inactivating the NF-κB pathway. Therefore, these results suggested that Quercetin can be used to treat AR [45]. Beyond nutraceuticals, several other adjuvant therapies are being explored to enhance the management of allergic rhinitis by modulating the immune response and reducing inflammation. Among these, probiotics have gained increasing attention for their potential immunomodulatory effects, with recent studies suggesting improvements in symptom control and quality of life in patients with allergic rhinitis. A systematic review and meta-analysis concluded that probiotics might improve symptoms and quality of life in AR patients, though the evidence is limited due to study heterogeneity [46]. Another review highlighted the positive effects of probiotic supplementation in treating AR, noting improvements in quality of life and modulation of the inflammatory response [47]. Furthermore, a meta-analysis focusing on pediatric patients demonstrated that probiotics effectively and safely improved AR symptoms and quality of life, though they did not prevent the onset of AR [48]. Recent findings also suggest that probiotics may reduce inflammatory markers and improve symptom control by shifting the immune balance towards a more tolerogenic profile [49]. All these findings highlight the growing interest in complementary approaches, such as probiotics and nutraceuticals, and several adjuvant therapies in optimizing allergic rhinitis management. While probiotics have demonstrated potential immunomodulatory effects, our study provides further objective evidence supporting the role of Quertal® as an effective add-on therapy for pediatric allergic rhinitis, showing significant benefits in reducing nasal inflammation and improving symptom control.
- 5.1. Strength of This Study
The strengths of this work are several. First, it is a unique study conducted in pediatric age that analyzes children affected by seasonal AR, monosensitised to grass pollens from a laboratory (eosinophils, tIgE, and sIgE for Phlp1), functional (nasal and exhaled nitric oxide, nasal cytology), subjective (NSS), and objective (AAR, spirometry) perspective. Furthermore, this work, in light of the encouraging results obtained, underlines the valuable
role of specifically formulated nutraceuticals as an add-on therapy for improving AR symptoms with a high safety profile. Lastly, the sample size is sufficiently large to make possible a valid statistical analysis. The findings of this study are particularly relevant to pediatric patients with mild-to-moderate allergic rhinitis who are monosensitized to grass pollen. However, caution should be taken when generalizing these results to populations with multiple sensitizations or severe disease requiring corticosteroids. Future studies should explore whether these benefits extend to broader populations, including adults or patients with more severe allergic profiles.
- 5.2. Limitations
- 5.3. Future Research Directions
To build on the findings of this study, future research should focus on larger cohorts and more extended treatment periods to fully assess the long-term effects of Quertal®. Expanding this study to include different populations, such as adults or patients with more severe AR, would provide more comprehensive data on its effectiveness across a broader range of patients. Additionally, conducting a placebo-controlled trial would provide more robust evidence of the true benefit of Quertal® in managing AR.
Future studies could also investigate the potential role of Quertal® as part of a maintenance therapy strategy to prevent exacerbations during peak pollen seasons. Assessing its use in conjunction with allergen-specific immunotherapy (AIT) or its impact on delaying the onset of asthma in pediatric patients with AR could offer new preventive strategies in allergy management.
6. Conclusions
This study highlighted that the nutraceutical Quertal®, used in addition to antihistamine therapy in a cohort of pediatric patients affected by AR, appears to induce significant benefits in nasal obstruction, reducing symptoms and alleviating local allergic inflammation from a subjective, objective, and functional point of view.
From a clinical perspective, this combined approach may benefit children with moderate symptoms who are not fully managed by antihistamines alone. One important implication for clinical practice is the potential to reduce the need for higher doses of
antihistamines, thereby minimizing side effects commonly associated with such therapies, such as drowsiness and cognitive impairment.
Additionally, Quertal®’s favorable safety profile makes it a compelling option for long-term use in pediatric patients. While the three-month treatment period provided encouraging results, extending therapy through peak pollen seasons or over a year could offer more significant insights into its prophylactic potential. Future clinical guidelines might consider incorporating nutraceuticals for long-term management, especially in patients with mild to moderate AR.
Given the chronic nature of AR and the potential for symptom persistence into adulthood, using a nutraceutical that offers both anti-inflammatory and immunomodulatory effects may help reduce the progression of allergic symptoms and possibly delay or prevent the onset of comorbid conditions like asthma.
Supplementary Materials: The following supporting information can be downloaded at: https: //www.mdpi.com/article/10.3390/jcm14051517/s1, Table S1: Consolidated Standards of Reporting Trials (CONSORT) completed checklist [50].
Author Contributions: A.G. and G.B. contributed equally to this work. A.M.Z. contributed to the conception and design of this study. G.B., A.G., A.M.Z., C.A. and A.S. contributed to the acquisition of data. G.B. performed the statistical analysis of data under the supervision of A.M.Z., G.B., A.G. and A.M.Z. contributed to the interpretation of the results and in drafting the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement: This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (or Ethics Committee) of the Sapienza University of Rome, Rome (0441/2023, approved on 23 November 2022).
Informed Consent Statement: Written informed consent has been obtained from the patient(s) to publish this paper.
Data Availability Statement: The original contributions presented in this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author(s).
Conflicts of Interest: The authors declare no conflicts of interest.
Figures
Used In Evidence Reviews
Similar Papers
Molecules (Basel, Switzerland) · 2016
Quercetin and Its Anti-Allergic Immune Response.
The Journal of allergy and clinical immunology · 2009
Complementary and alternative medicine: herbs, phytochemicals and vitamins and their immunologic effects.
Allergy, asthma, and clinical immunology : official journal of the Canadian Society of Allergy and Clinical Immunology · 2020
Quercetin with the potential effect on allergic diseases.
Autoimmunity · 2023
Quercetin improves the imbalance of Th1/Th2 cells and Treg/Th17 cells to attenuate allergic rhinitis.
Primary care · 2002
Respiratory and allergic diseases: from upper respiratory tract infections to asthma.
Phytomedicine : international journal of phytotherapy and phytopharmacology · 2004